

The use of innovative adhesive technologies allows manufacturers to capitalize on the advantages that composites offer.
Metal is one of the most common substrates associated with adhesives in the industrial market. Automobiles, heavy trucks, freight trailers, agricultural, and heavy equipment have all traditionally been major users of steel, aluminum, and other metals and alloys. As the requirements of these vehicles have grown over time, however, the metals in many applications are slowly being replaced by composite structures. This trend, in turn, affects the adhesives industry.

Composites are used extensively in trailer manufacturing.
Environmental Pressures and Composites
As the focus on environmental awareness and preservation intensifies, the transportation industry is increasingly affected by government regulation and general public sentiment. The result of these pressures has been a significant effort by OEMs (especially those in the automotive and heavy truck/trailer industry) to reduce emissions and improve vehicle gas mileage. These efforts have led to the use of unique substrates, specifically composites, to help decrease vehicle weight and therefore improve gas mileage.With many of these substrates, mechanical fasteners have been abandoned in favor of adhesives, which provide a two-fold advantage. First, they provide a process and design flexibility that mechanical fasteners simply do not. Second, the adhesives themselves are lighter in weight than mechanical fasteners such as rivets, especially in large-quantity uses such as trailer sidewalls. In addition, adhesives can provide a cost savings over mechanical fasteners.
Composites are used extensively in vehicle and trailer manufacturing. Some applications include:
- Automotive hoods, doors and deck lids
- Heavy truck hood, roof and door assemblies
- Trailer and storage box sidewalls and roofs
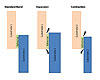
Figure 1. Stress Caused by Differing Coefficients of Thermal Expansion
Surface Variation
Each composite surface can vary in terms of roughness, porosity or surface energy. In some cases, a coating may have been applied prior to bonding that completely changes the chemistry of the part surface. The physical and chemical attributes of the bonding surface have a huge influence on the adhesion strength of the bond. In general, high surface area substrates are easier to bond to. However, extremely porous materials can be a problem if the adhesive is forced into a non-continuous form.Surface energy is even more critical to the successful bonding of a set of substrates. For optimum adhesion, an adhesive must be able to fully “wet” the substrate surface. This occurs when the adhesive is lower in surface energy than the substrate. Table 1 contains surface energies of several common substrates and related materials. The information in the table shows that metals are significantly higher in surface energy than plastics. But the line between what is generally considered “high surface energy” and “low surface energy” is fairly narrow-about 10 mJ/m3. Developing an adhesive that bonds to these very different surfaces (metal vs. plastic) can require significantly different approaches and materials.
To complicate matters further, in situ mold release is commonly used to prevent molded composite parts from “sticking” in the mold. The mold release is formulated to migrate to the surface of the part during molding, where it can prevent the part from adhering to the mold. This mold release is still present on the surface of the composite part to varying degrees when it is bonded. Adhesive manufacturers must formulate a product that is able to adhere to the surface of the part when it contains a mold release specifically designed to prevent adhesion.
Formulating an adhesive to overcome this scenario is no small task. The use of a selective wetting agent based on the specific chemistries involved provides the best opportunity to adequately bond the part, especially if the application calls for environmental cycling resistance. However, it is critical to note that each application is different, and one solution may not work for every part or process.
Further surface variation can come from parts that experience deviations in the molding process. Differences in mold time, temperature and pressure can affect the porosity of the part, as well as the surface energy. Parts can also be molded in color, which requires that a part contains a color pigment. One common issue with molded in color parts is marring. To avoid this problem, substrate manufacturers include an anti-marring agent such as PTFE wax. This wax orients at the surface and acts like a low-surface-energy coating that the adhesive must penetrate to gain adhesion.
Adhesion difficulties due to surface effects and treatments can be present in initial adhesion testing such as lap shear or T-peel. However, it may also be hidden in these tests only to resurface during more demanding adhesion tests, such as hot water immersion (90-100°C) or environmental cycling. When bonding composites in combination with more traditional substrates, it is important to perform a variety of tests to verify results.

Bonding Dissimilar Substrates
Bonding two surfaces made from the same material is straightforward. Bonding two parts made from disparate materials, however, can be a challenge. First, the aforementioned factors affect the longevity of the bond. The more the two surfaces vary, the more difficult it can be to bond them together, as the adhesive must have a broad enough set of adhesion properties to be able to match up with both parts. The coefficient of thermal expansion (the degree to which a substrate expands and contracts with heat or cold) is also of critical importance when bonding dissimilar substrates. This is particularly true with bonding composites to rigid substrates like metals.Metals are typically higher in coefficient of thermal expansion than composites, and this difference can be significant. When a metal substrate expands or contracts at a higher rate than the more thermally stable composite, it creates a stress in the adhesive bond as it “stretches.” In the worst case, the bond, unable to tolerate the stretching effect, will break and the adhesive will pull free from the metal, causing adhesive failure. In less dramatic situations, the adhesive may not break away immediately, but constant fluctuations in temperature may cause repeated stresses in the bond and potentially result in fatigue and adhesion loss.
It is critical that the adhesive is formulated to take these cyclical stresses into account and tested to verify efficacy. Figure 1 illustrates the stresses created when one substrate has a higher thermal expansion coefficient. In the illustration, substrate 1 is the more thermally stable substrate (composite), and substrate 2 has the higher coefficient of thermal expansion, resulting in a higher degree of expansion or contraction.
In addition to the stresses caused by expansion and contraction, fracture toughness must be considered. Impact is a serious concern, especially in the transportation industry. The bond must be able to survive substantial stress from impact without fracture, which would result in adhesive failure. Fracture tests are commonly performed at ambient conditions as well as in extreme cold (e.g., from -20°C to as cold as -40°C). At these extreme temperatures, many adhesives fracture upon impact. The root issue in this scenario is that the adhesive becomes too brittle to absorb and efficiently displace the energy of the impact.
Adhesive manufacturers have made significant efforts over the last several years to develop toughened systems that can pass these cold impact tests. The first step toward meeting this requirement is fine-tuning the curing process of the adhesive to avoid brittleness and improve fracture toughness. The use of specially formulated accelerators can help adjust the cure rate and improve toughness.
A second critical formulation area is the use of selective, reactive tougheners in combination with thermoset polymers, which results in a highly toughened adhesive that does not sacrifice mechanical properties. These avenues are far superior to the traditional method of “softening” the polymer matrix with thermoplastic materials that will degrade mechanical properties. Further development in this area continues.
Overcoming Obstacles
It is critical to spend early development time using failure mode and effects analysis (FMEA) and establishing a design plan that minimizes or eliminates the potential failure modes identified as part of the design FMEA. Using multiple tests to evaluate failure modes is also a key component to success.Once design and test strategies are established, development can begin. Optimizing polymer type and functionality, additive levels and toughening levels are critical components to reaching the best formulation possible. However, consideration of the customer’s manufacturing process must be given as well. Pairing the adhesive and the application process, including optimum cure parameters, provides the greatest chance of long-term success.
Composites provide significant advantages in various applications, especially in the transportation industry. The use of innovative adhesive technologies allows manufacturers to capitalize on the advantages that composites offer. However, composite applications are among the most challenging developments an adhesives manufacturer can undertake. Many obstacles to product success exist, and the developer must be cognizant of the potential issues related to surface profile and energy, variations in molding process, surface coatings, or contaminants.
When bonding dissimilar substrates, consideration must be given to the differences in the substrates, including coefficient of thermal expansion and fracture toughness. Finally, pairing the adhesive with the manufacturing process helps ensure the most optimum results possible.
For more information, contact the author at Uniseal, Inc., 1014 Uhlhorn St., Evansville, IN; phone (812) 425-1361; email bwillis@uniseal.com; or visit www.uniseal.com.
The author would like to thank Thanikaivelan Veeraraghavan, Ph.D., for his contributions.

