Carton closure hot-melt packaging adhesives using ethylene vinyl acetate (EVA) polymer systems have been used for decades due to their availability, formulation latitude and performance attributes.1 Packaging adhesive formulations typically contain polymer, tackifier resin and wax.
Packaging adhesives using EVA can be formulated using a variety of tackifier systems, including rosin esters, hydrocarbon resins (C5, C9, C5/C9), terpene resins and hydrogenated hydrocarbon resins.2-7 However, for premium packaging adhesive applications where clarity, thermal stability and temperature resistance are critical, EVA-based packaging adhesives are often formulated with hydrogenated hydrocarbon tackifiers.
The hot-melt adhesive industry has recently trended toward high-quality packaging adhesives due to cost benefits and improved performance characteristics. This has prompted the development of aromatic pure monomer resins (PMRs) as synergetic tackifier additives for EVA-based premium packaging adhesives.
PMRs are produced from pure aromatic monomer streams, especially styrene and alkyl styrene derivatives. Compared to other aromatic crude C-9 resins, major advantages of PMRs include light color (water white), excellent thermal stability, temperature resistance (similar to hydrogenated hydrocarbon resins) and controlled molecular weight.8-14
In this study, the effect of different PMRs and their benefits in performance attributes when used in premium EVA hot-melt packaging adhesives are described. It was determined that packaging adhesives containing PMRs can effectively improve adhesive and cohesive performance characteristics while maintaining other performance attributes of the adhesive. PMR-containing adhesives also exhibit good thermal stability and temperature resistance.
Results and Discussion
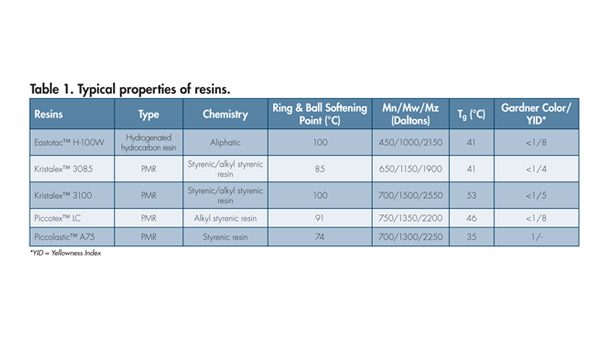
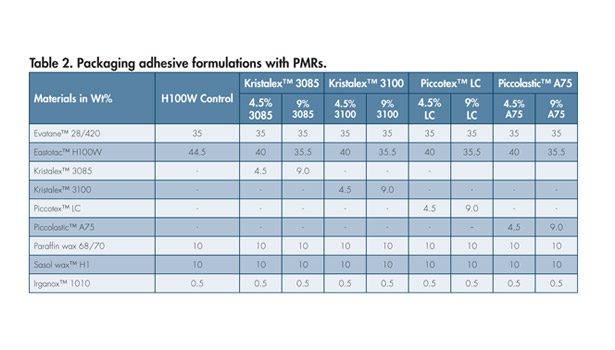
Typical properties of selected hydrogenated hydrocarbon resin and PMRs are given in Table 1. Values shown are an average of typical samples and should not be interpreted as product specifications. For this study, a packaging adhesive control formulation was prepared with a hydrogenated hydrocarbon resin.* Four different PMRs with hydrogenated hydrocarbon resins were evaluated at two different addition levels (4.5 and 9 wt %). Table 2 shows the formulations used in the study.
These formulations were prepared using an in-can mixer with mechanical agitation (a paddle-type agitator controlled by a variable speed motor) in pint-sized cans with a heat block set at 177°C. The polymer and antioxidants were introduced into the can and heated up to 177°C under a nitrogen blanket. Resin followed by wax was then introduced into the can after the polymer was melted. This was agitated for 30 min until the mixture was completely homogenous.
Viscoelastic characteristics of PMR-containing formulations were evaluated using dynamic mechanical analysis (DMA), as shown in Figure 1. A TA Instruments Ares RDA3 rheometer was used in a parallel plate geometry (8 mm plate, 1.704 mm gap) and autostrain mode (maximum strain at 5.0 %) at 10 Hz frequency with a heating rate of 6°C/min.
As seen in Figure 1, elastic modulus is not affected by the PMR addition and shows similar viscoelastic characteristics as the control formulated with only resin (without PMR). Even though PMR-containing formulations show a slightly higher glass-transition temperature (Tg), the Tg profiles are very similar to control with no PMR.
Figure 2 shows the shear adhesion failure temperature (SAFT) and peel adhesion failure temperature (PAFT) on 40-lb virgin Kraft paper for the PMR-extended formulations along with hydrogenated hydrocarbon resin control. Significant improvement in SAFT and PAFT was observed for 9 wt% PMR-containing packaging adhesives, while 4.5 wt% PMR-containing formulations show slight improvement in PAFT and similar SAFT performance compared to control with no PMR.
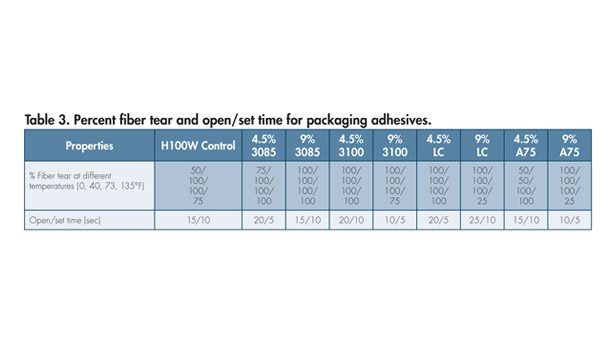
Table 3 shows corrugated-to-corrugated cardboard adhesion fiber tear at various temperatures and open/set time characteristics of packaging adhesive formulations measured using an Olinger testing equipment (in five-second intervals). Samples were prepared using the Olinger at 300°F application temperature with an adhesive coating weight of 0.15-0.25 g on a 2 x 2.5 corrugated substrate.
PMR addition shows improvement in cardboard fiber tear adhesion characteristics, with Kristalex 3085 being the best. It can be seen from Table 2 that the PMR amount does not affect the adhesive set time. However, PMRs have definite influence on adhesive open time. Viscosity profiles at three different temperatures for PMR-containing formulations are very comparable to controls (without PMR) even after aging at 177°C for 96 hrs (see Figure 3).
Figure 4 shows pictures of the formulations before and after aging. Aging was performed in an air-drafted oven at 177°C for 96 hrs. The before aging and after aging characteristics for the PMR-containing formulations are similar to or better than the control formulation.
Summary and Conclusions
PMRs can be used as an effective synergistic tackifier for premium EVA packaging adhesives, where improved adhesive properties and thermal stability characteristics are essential. PMRs improve adhesive properties such as SAFT, PAFT and adhesion (fiber tear) at different temperatures. They have similar viscoelastic characteristics when compared to control formulations with no PMR. Improved open/set times were also observed. Finally, PMRs display a good thermal stability upon aging, as well as good color and viscosity stability. The use of PMRs and hydrogenated hydrocarbon resins with EVA polymers is a cost-effective solution for premium packaging adhesives technology in today’s environment, where improved thermal stability, clean runnability and adhesive performance characteristics are critical.
For more information, call (800) 327-8626, email adhesives@eastman.com or visit www.tackifier.com.
Acknowledgements
The author would like to thank Alicia Powers, Christopher Jones, Timothy Williams, Andrea Hagood and Deborah Moroney of Eastman Chemical Co. for their support in obtaining data.
References
1. Schlademan, J.A., Adhesives Age, Aug. 1975, 45.
2. Honiball, W.J., Adhesives Age, May 1994, 21.
3. Godfrey, D.A., Ardemagni, L.A., Tappi Journal, 1994, 77, 88.
4. McBride, E., Adhesives Age, May 1995, 22.
5. Honiball, W.J., Lebez, J., Simons, J.C., Tappi Journal, 1997, 80, 179.
6. Lecointe, J.P., Magny, B., Langlet, J., Badie, P., Adhesives Age, Nov. 1999, 27.
7. Barrueso-Martinez, M.L., Ferrandiz-Gomez, T.D.P., Martin-Martinez, J.M., Adhesives Age, Apr. 2001, 32.
8. Holohan Jr., J.F., Penn, J.Y., Vredenburgh, W.A., Kirk-Othmer Encyclopedia of Chemical Technology; Wiley InterScience, New York, 1980, 3rd Ed., Vol. 12, pp. 852-869.
9. Collin, G., Mildenberg, R., Zander, M., Ullmann’s Encyclopedia of Industrial Chemistry; Wiley-VCH Verlag Gmbh & Co.: Weinheim, 2003, 6th Ed., Vol. 31, pp. 357-369.
10. Findlay, J., Bikales, N.M., Encyclopedia of Polymer Science and Technology; John Wiley & Sons, New York, 1968, Vol. 9, p. 853.
11. Powers, P.O., U.S. Patent No. 3000868, 1961.
12. Campbell, C.C., Finfinger, D.A., U.S. Patent No. 3630981, 1971.
13. Douglas, P.S., Patellis, A.P., Vredenburgh, W.A., U.S. Patent No. 3932332, 1976.
14. Campbell, C.C., Finfinger, D.A., U.S. Patent No. 3956250, 1976.