

Even with a greatly variable molecular structure, epoxies generally share adhesion to a wide range of substrates, resistance to a wide range of chemicals and excellent mechanical properties. Modification of epoxy-resin systems by additives, modifiers, fillers, plasticizers and other polymers offers vast opportunities to further customize the attributes of these materials. These positive attributes and easily varied structure allow formulators the opportunity to tailor a resin system's handling, cure behavior and cured performance over a tremendous range.

Elastomer-Modified vs. Flexibilized Epoxies
Today, elastomer-modified epoxy resins play a critical role in adhesives and damage-resistant coatings. The toughening mechanisms of elastomer-modified epoxy systems are different from flexibilized epoxy systems and can be used in combination with them. Flexibilized epoxy systems reduce mechanical damage through lowering modulus or plasticization; this allows stress to be relieved through distortion of the adhesive1,2,3.In contrast to flexibilized systems, elastomer-toughened epoxy systems generally maintain a large percentage of the modulus and temperature resistance of the unmodified resin system. They relieve stress by cavitation of the elastomer particles and shear banding in the cavitated zone4,5,6,7,8. Elastomer-toughened epoxy systems tolerate damage by preventing crack growth and thus limiting the damaged area.
In practice, elastomer concentrations of 5% to 15% by weight are commonly used. The addition of elastomers to epoxy adhesives can also improve peel strength, fracture resistance, adhesion to oily surfaces and ductility.
Considerable work has shown that the separation of the elastomer into a separate phase is necessary for significant toughening to occur and to retain the bulk properties of the epoxy system5,7,10. Thus, particle size and distribution of the elastomer phase is very important.
To obtain the desired phase separation and particle-size distribution, functionalized elastomers are usually pre-reacted (adducted) to a base epoxy resin. Adduction reduces the likelihood of early phase separation and maintains the solubility of the elastomer in the uncured resin system by increasing the molecular weight of the elastomer and by changing the solubility parameter of the elastomer to a value between the unreacted elastomer and the base epoxy resin.
The primary investigated elastomers have been functionalized butadiene-acrylonitrile copolymers. These elastomers are available with epoxy-terminated (ETBN), carboxy-terminated (CTBN), amino-terminated (ATBN) and vinyl-terminated (VTBN) functionalities6,7,9.
CTBN have been the elastomers of choice because of their miscibility in many epoxy resins, the ease with which an adduct to the epoxy resin can be formed (acid-epoxy reactions can be accomplished very cleanly) and the stability of the resulting adducts. It is important to completely react all the carboxylic acid groups to have this consistency of behavior. If a CTBN is not pre-reacted with an epoxy resin, the carboxylic acid groups can react with the curing agent, the substrate or not at all; these change the solubility of the elastomer and affect the degree of separation from the bulk resin system and the particle size.
One of the limitations of epoxy-CTBN adducts has been their high viscosity, which limits the types and quantities of additional additives that can be used by formulators. We have recently commercialized a new epoxy-CTBN adduct, EPONR Resin 58003, and have a second developmental adduct, Epoxy Research Resin RSM-2577, which have significantly lower viscosities compared to industry-standard BPA-based epoxy-CTBN adducts. BPA stands for the bisphenol of acetone. BPA is the primary building block of the most widely used commercial epoxy resins.
In addition, lower concentrations of these new epoxy-CTBN adducts are generally needed to achieve equivalent adhesive performance. As a result, epoxy resins modified with these new epoxy-CTBN adducts can have significantly lower viscosities, which provide formulators more opportunities to vary the types and quantities of additional components in their adhesive systems.
Experimental
This article compares the lap shear strengths of adhesive systems modified with the new low-viscosity epoxy-CTBN adducts (Low Vis CTBNs) and a standard BPA Epoxy-CTBN adduct (BPA CTBN-H). Two liquid epoxy resins were used as a basis for these adhesive systems. One is an industry-standard epoxy based on the bisphenol of acetone (BPA Epoxy), and the other is a lower viscosity, undiluted epoxy based on the bisphenol of formaldehyde (BPF Epoxy). Table A describes the epoxy resins and epoxy-CTBN adducts used in this study.The high-acrylonitrile CTBN used in EPON Resin 58005 (BPA CTBN-H) and EPON Resin 58003 (Low Vis CTBN-H) makes these resins compatible with most epoxy-resin types, including BPF epoxies and novolac epoxies, within the typical-use range of less than 50% by weight. The moderate-acrylonitrile-level CTBN used in Epoxy Research Resin RSM-2577 (Low Vis CTBN-M) generally restricts this resin to compatibility with BPA Epoxy resins within the typical-use range of less than 50% by weight.
Two common room-temperature curing agents were used, EPI-CURER Curing Agent 3072 (amidoamine) and EPI-CURE Curing Agent 3140 (polyamide). The filler used for this study was Vicron 25-11, a ground calcium carbonate. The amount of additional filler achievable in adhesive systems using the new Low Vis CTBNs is quantified for a limited number of systems to demonstrate the wider formulation possibilities and potential cost effectiveness. (See Table B, page 31)
Sample Preparation
Freshly acid-etched 2024-T3 aluminum coupons were the adherends for the lap-shear tests. The coupons were 1" x 4" x 0.030" and were etched for approximately 0.75" on one end. Preparation and etching were done per ASTM D-2651-90, Standard Guide for Preparation of Metal Surfaces for Adhesive Bonding. As called for in the guide, a P-2 etch was used on the aluminum. Etched aluminum coupons were stored in n-hexane.Stoichiometric amounts of the amidoamine and polyamide were used. Resin systems were mixed just prior to use and applied at room temperature. The resin system was applied to one side of the etched surface of two coupons with a tongue blade to approximately 0.010" thick. Two 0.015" nylon monofilament spacers were placed in the bonding area, parallel to the long dimension and equally spaced laterally. The coupons were assembled with a 0.50" lap and clamped with small binder clamps to maintain orientation during cure. Specimens were cured at room temperature (23degC) for seven days.
The physical properties of the cured resin systems were measured on neat resin castings. These were prepared by casting the resin system in molds consisting of plate-glass sheets separated by a 1/8-inch spacer. The castings were cured for seven days at room temperature before cutting into specimens and testing.
Lap-shear tests were performed according to ASTM 1002 using a crosshead speed of 0.050 inch/minute. Tensile tests were performed according to ASTM D638 using a crosshead speed of 0.200 inch/minute. Test conditions for the ASTM tests were 23degC and 50% relative humidity. Glass-transition temperatures (Tgs) were determined using a differential scanning calorimeter (DSC) with a heating rate of 20degC/min.

Results and Discussion
Lap-Shear and Casting Properties
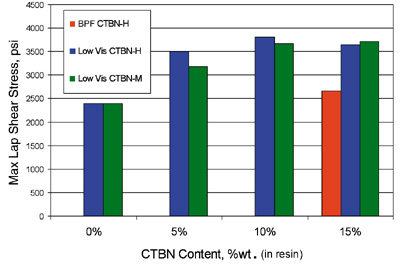
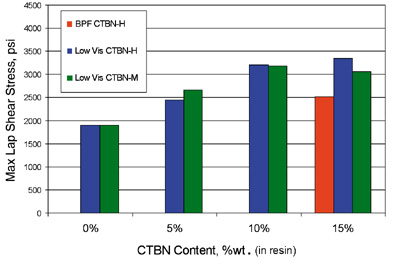
Lap-shear strengths of the three epoxy-CTBN adducts blended with the standard BPA Epoxy are shown in Figures 1 and 2. Lap-shear performance of adhesive systems containing 5% elastomer using either Low Vis CTBN-H or Low Vis CTBN-M are equivalent or slightly better than adhesive systems containing 15% elastomer using the standard BPA CTBN-H. In these adhesive systems, Low Vis CTBN-H is generally superior to Low Vis CTBN-M.Figures 3 and 4 show lap-shear strengths of the three epoxy-CTBN adducts blended with the BPF Epoxy. In this series of tests, the lap-shear strengths of the unmodified BPF Epoxy systems were notably higher than those of the BPA Epoxy systems.
The trend of improved strength with the Low Vis CTBNs is similar to that observed with the BPA Epoxy systems. However, observed improvements with the Low Vis CTBNs are less dramatic. Again, the lap-shear performance of systems containing 5% elastomer using either Low Vis CTBN-H or Low Vis CTBN-M are equivalent to, or slightly better than, systems containing standard BPA CTBN-H at 15%-elastomer concentration.
Tables C and D list the physical properties of cured castings made from the CTBN-modified BPA Epoxy systems. As expected, tensile strength and modulus properties are reduced by increasing CTBN content, roughly in proportion to the amount of the epoxy matrix replaced by the CTBN.
As previously mentioned, one of the advantages of elastomer-toughened epoxy adhesives is the minimal impact on elevated-temperature performance. The Tgs of these systems fall only slightly, indicating efficient phase separation of the CTBN elastomer during cure. If the CTBN were not separating well from the epoxy during cure, one would see a large drop in the Tg.
Viscosity, Filler Loadings and Cost Comparisons
Based on lap-shear results, the new Low Vis CTBNs blended with either the BPA Epoxy or the BPF Epoxy provide equivalent lap-shear strengths at lower elastomer concentrations than the current standard BPA CTBN-H. This, plus their lower initial viscosities, results in substantially lower resin-blend viscosities, which can provide more opportunities for formulators to use additional fillers, modifiers, etc. to tailor the properties of the final formulated adhesives.As shown in Figure 1, the lap-shear strength of the 5%-wt. elastomer-content Low Vis CTBN-H in BPA Epoxy (New System) is equal to or better than the 15%-wt. elastomer-content BPA CTBN-H in BPA Epoxy (Standard System). The 25degC viscosity of the Standard System is approximately 85,800 centipoise (cP) while the New System is only 22,400 cP. Figure 5 plots the effect of rubber concentration on viscosity for Low Vis CTBN-H and BPA CTBN-H blended into both BPA and BPF Epoxies.
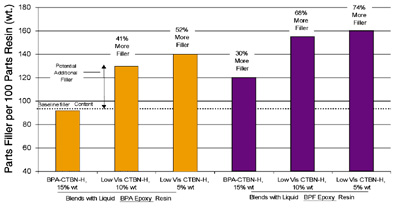
The amount of a typical inert filler (ground calcium carbonate) necessary to reach a paste viscosity of 500,000 cP was determined to demonstrate the effect this viscosity difference might have on the quantity of additional filler and the resulting formulated cost. The filler contents of these and four other systems are shown in Figure 6. The New System could accommodate additional filler equal to approximately 45% of the unfilled resin-system weight compared to the Standard System. If we carry this example one step farther, relative system raw-material costs can be calculated.
New System Can Reduce Costs
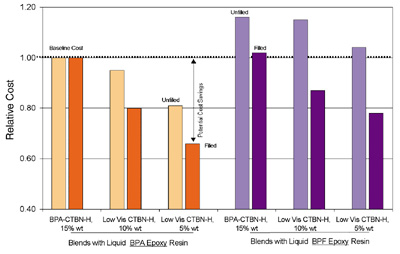
Figure 7 plots relative costs of six adhesive systems with and without calcium carbonate filler. Cost calculations were based on published prices.As indicated, the costs of systems that use the new Low Vis CTBNs can be cost advantaged for equivalent adhesive strength. Compared to the Standard System, the New System cost would be approximately 19% lower without filler and approximately 34% lower with filler.
Although the example systems are very simple, they have been used to demonstrate that these new Low Vis CTBNs provide the option to use higher additive levels that can be used to tailor adhesive properties to meet specific end-use requirements at competitive costs. It is also interesting to note that a very low viscosity (~8,000 cP without filler), cost-effective adhesive system can be obtained by blending BPF Epoxy resin with Low Vis CTBN-H. (See Figures 3, 5 and 7.)
Conclusions
The two new elastomer-modified epoxy tougheners evaluated in this study are lower in viscosity and provide improved lap-shear strengths compared to standard elastomer-modified liquid BPA Epoxy tougheners. The improved shear strengths allow the opportunity to formulate higher performance adhesives or to use reduced amounts of toughener to achieve equivalent performance. The lower initial viscosity plus the reduced elastomer content result in significantly lower resin-system viscosity. This allows formulators more latitude to adjust system costs and/or performance through the use of additional fillers and modifiers.This article is based on a paper presented at the October 2000 Adhesive and Sealant Council Fall Convention. Additional information on EPONR Epoxy Resins and EPI-CURER Curing Agents is available from Resolution Performance Products LLC (formerly Shell Epoxy Resins Business), PO Box 4500, Houston, TX 77210-4500; call 1-800-TEC-EPON (1-800-832-3766); or visit the Web site www.resins.com. Or Circle No. 67.
References
1 Farrakhov, A. G. and V. G. Khozin, Mechanical behavior of plasticized epoxy polymers, pp. 585-597, Crosslinked Epoxies, B. Sadlacek and J. Kahovec editors, Walter de Gruyter and Co., Berlin, 1987.2 Misra, S. C., J. A. Manson, and L. H. Sperling, Effect of cross-link density distribution on the engineering behavior of epoxies, pp. 137-156, Epoxy Resin Chemistry, ACS Symposium Series 114, Ronald S. Bauer, editor, American Chemical Society, 1979.
3 Ellis, Bryan, Chemistry and Technology of Epoxy Resins, pp. 225-228, Blackie Academic and Professional, London, 1993.
4 Ellis, Bryan, Chemistry and Technology of Epoxy Resins, pp. 131-138, 229-233, Blackie Academic and Professional, London, 1993.
5 Krasheninnikov, A.I., A.M. Ternovykh, M.O. Slobodchikov, and V.S. Karentnikov, Influence of phase structure on the mechanical properties of rubber-modified epoxies, pp. 443-450, Crosslinked Epoxies, B. Sadlacek and J. Kahovec editors, Walter de Gruyter and Co., Berlin, 1987.
6 Bagheri, Reza and Raymond A. Pearson, The role of cavitation in rubber-toughened epoxies, International SAMPE Technical Conference 25, 1993, pp. 25-39.
7 Daghyani, H. R., Lin Ye, Yiu-Wing Mai, and Jingshen Wu, Fracture behaviour of a rubber-modified tough epoxy system, Journal of Materials Science Letters 13 (1994) 1330-1333.
8 Maazouz, A., H. Sautereau, and J. F. Gerard, Toughening of epoxy networks using pre-formed core-shell particles or reactive rubbers, Polymer Bulletin 33, 67-74 (1994).
9 Verchere, Didler, Henry Sautereau, Jean-Pierre Pascault, S.M. Moschiar, C.C. Riccardi, and R.J.J. Williams, Rubber-modified epoxies, Advances in Chemistry Series 1993 Vol. 223, The American Chemical Society.
10 Karger-Kocsis, J. and K. Friedrich, Microstructure-related fracture toughness and fatigue crack growth behavior in toughened, anhydride-cured epoxy resins, Composites Science and Technology 48 (1993) 263-272.
