Transposable Adhesives: Acrylic Pressure-Sensitive Adhesives with Reactive Components
This paper was awarded the Pressure Sensitive Tape Council’s 2013 Carl A. Dahlquist Award for Best Technical Paper.



Pressure-sensitive adhesives (PSAs) have been an integral part of many industries that require the bonding of two or more materials. PSAs are commercially
| Jump to: |
available in many forms: hot/warm melts, solvent borne, water based and syrups. They are used for applications as simple as paper labels to high-performance tapes used to bond two pieces of an automobile. Regardless of the application, PSAs have a set of viscoelastic properties that exhibit permanent adhesion characteristics once applied to a substrate. These adhesion characteristics are constant outside the typical responses from degradation from external environmental conditions, such as temperature and chemical exposure.
With advancements in technology over the past decade, applications now exist that require PSAs to show different adhesion performances at different stages of the application process. An example would be a PSA that exhibits properties like a removable label at application, but transposes to a high-strength PSA similar to an HVAC tape in a second stage, when activated by an external stimuli. This concept extends beyond just transposition from one PSA state to another; it can also include transpositions from a PSA state to a structural bond. Additional uses include the need for transposition from high adhesion to low adhesion (debond on demand) that has been heavily researched and well documented.2-6
PSAs can transpose from low adhesion properties to high adhesion properties via precise thermal- or UV-activated chemistries. For structural adhesive tape applications, improved tape stability enables the adhesive to be supplied in tape form for easy and efficient application through controlled bond lines, which is an advantage over the less-favorable approach of using applicator guns.
Background
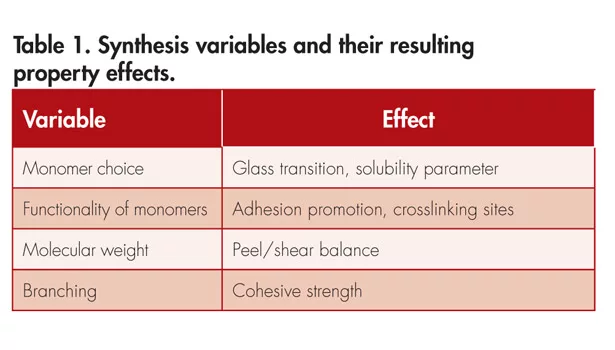
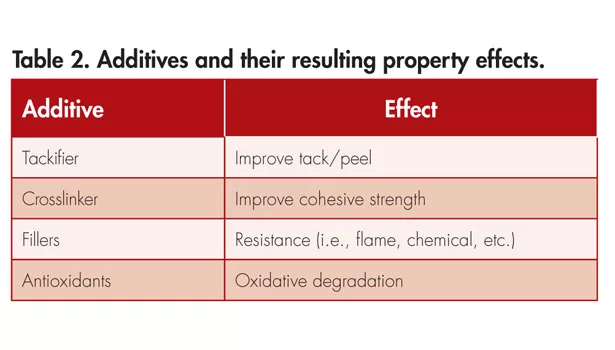
The adhesion properties of a PSA can be manipulated by multiple variables during the synthesis of the polymer. In Table 1, a few major contributing variables, along with their property effects, are displayed. In addition to synthetic variables, additives are introduced to the polymer post-synthesis to help enhance certain adhesion properties of the PSA. Some examples of these additives are shown in Table 2.
The adhesion properties of a PSA can be manipulated by multiple variables during the synthesis of the polymer. |
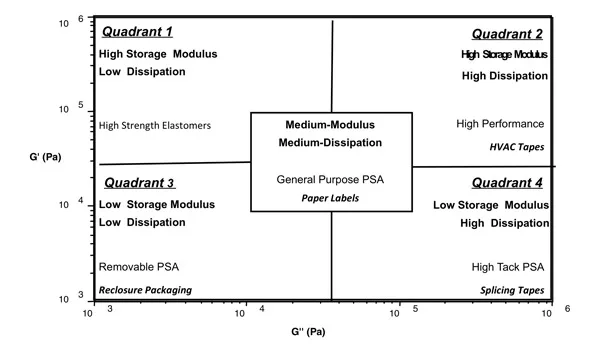
With the combination of the synthetic variables and additives, a broad range of PSA performance can be achieved. Performance can be qualified by the viscoelastic properties of the PSA.7 Dynamic mechanical analysis (DMA) enables the industry to measure the viscoelastic properties of PSAs. In this technique, the storage (G’) and loss (G”) moduli are measured at two frequencies that represent the bonding and debonding states. With the corresponding measurements, the PSA performance window can be predicted based on where it falls on the plot depicted in Figure 1. All PSAs will have a performance window somewhere in Figure 1.
Using this method of characterization, data will confirm the transposition of PSAs from one set of PSA performance to another (i.e., from one quadrant to another). Data will also suggest a PSA transposing from one PSA quadrant to a structural state located well above the Dahlquist criteria (G’=105).8
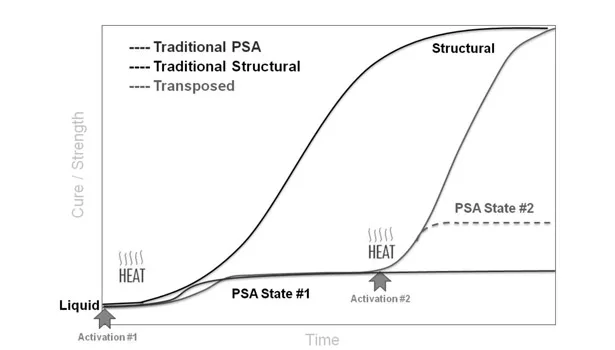
The goal of the following transposable adhesive example was to use a thermal or UV activator that would trigger a chemical reaction to cause the adhesion properties to change after application. Whether the change in adhesion properties was to another PSA type or to structural was dependent on the application. In these examples, the transposition chemistry is epoxy polymerization via the activators. Figure 2 shows the transposition process as a function of cure/strength vs. time.
Activation 1 designates the coating process or mixing of 100% solids, two-part structural adhesive, which yields a PSA of State 1 or structural state, respectively. At this point, general crosslinking present in the base polymer system is contributing to the PSA performance, but not the epoxy chemistry. Activation 2 occurs sometime after application and designates the triggerable reaction to cause the transposition to PSA State 2 or structural. After Activation 1, there is no change in Performance State 1 after application until Activation 2. In comparison, traditional PSA and structural adhesives have only one activation (coating/drying PSAs or mixing two-part structural adhesives) and directly reach their end state. Transposable adhesives allow the user to take advantage of PSA State 1 for extended periods of time and activate to State 2 on demand by either thermal or UV exposure.
Experimental
The same base solution polymer was used for all the examples, which differ only by the amount of epoxy diluent added. Base acrylic esters, such as 2-ethylhexyl acrylate (EHA), butyl acrylate (BA), and acrylic acid (AA), were obtained from various commercial suppliers and used as received to polymerize the base polymer. The acrylic polymerization was initiated with azobis(isobutyronitrile) (AIBN) and made in organic solvents. The base polymer was formulated with aluminum acetoacetonate (AAA) at various levels by weight based on polymer solids. Epoxy S-21 was obtained from Synasia and used as delivered. The thermal super acid generator was obtained from King Industries and used as received.
All samples were coated at approximately 2.0 mil adhesive thicknesses onto 100% solids, platinum-cured silicone paper liner. The coatings were all air dried for
10 minutes and placed in a forced air oven for 10 minutes at 80°C and closed with 100% solids, platinum-cured silicone paper liner. Adhesion testing was done on transfer coatings to 2.0 mil aluminum foil. The laminates were all aged in a controlled climate room (70°F at 50% humidity) for 24 hours prior to testing. The transposition was done via thermal activation of epoxy polymerization at 140°C for 15 minutes after application by the designated dwell time. The UV example was transposed via an Uvitron Intella-ray unit fitted with 600 W/in UV-B bulb. The adhesive was exposed to UV light and then immediately used to construct an aluminum lap shear such that the transposition chemistry was initiated but completed after the lap shear construction was made.
Dynamic mechanical analysis (DMA) was performed on a TA Instrument AR-2000 rheometer using parallel plate clamps. Samples of 1.0 mm thickness were placed in the clamp and equilibrated to 30°C. Frequency sweeps were conducted from 0.01-100.00 rad/s at 30°C to construct the viscoelastic windows. Temperature sweeps were conducted from -80-180°C to measure storage modulus at 10 rad/s.
Results and Discussion
The first example is shown in Table 3, where the adhesive system transposes from a low-strength adhesive to a structural end state via UV activation. The tensile strength is reported for a 1 x 1-in. overlapped aluminum lap shear (ASTM D 1002) for the base PSA, a high-performance PSA modified with reactive silane oligomer to form a high-strength interpenetrating network (IPN), and the base PSA transposed to structural.9
The lap shear data shows that the transposable adhesive system has a tensile strength that is 10 times that of the base adhesive, and it exceeds the tensile strength of a high-performance IPN used in industrial tapes (400 psi was the maximum for the load cell used in the study). In comparison, high-strength structural adhesives have a tensile strength of ~ 1000 psi (7 MPa); however, some applications only require up to ~ 300 psi (2 MPa) to create a structural bond.10
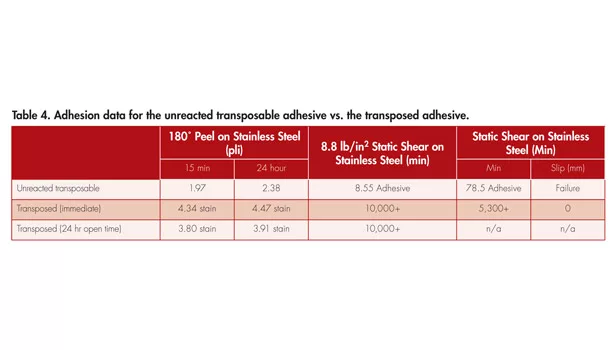
The next example used the same base polymer and is transposed to a high-performance type PSA. The 180° peel strength (ASTM D 3330) off stainless steel after a 15-min and 24-hr dwell is reported, along with room temperature (ASTM D 3654) and 65°C static shear, in Table 4.
As reported, the peel adhesion more than doubles after the thermal activation of the epoxy polymerization. Not only does the peel adhesion double, but the shear also dramatically increases. This is unique because typical PSAs have a balancing effect when talking about peel and shear. This effect shows that, as there is an increase in either peel or shear, the other property typically decreases. The enhancements of adhesion and cohesion properties that the transposable adhesive exhibits are also present when the open time (time after application but before epoxy activation) is changed from immediate to 24 hours. Similar adhesion data between these preactivated dwell times suggest this transposable tape system has long/stable open time. This property allows users to apply, remove and reapply the tape before activation, which makes a permanent bond at the user’s will. In addition to increased adhesion, temperature resistance increases with transposition and is supported by the 65°C shear data included in Table 4. Resistance to temperature makes these tapes attractive for automobile applications under the hood or any application where the adhesive has to withstand high temperatures without degrading.
For Example 2, the transposition was thermally activated at 140°C for 15 minutes. These conditions may not be acceptable for some applications. To find the limits of the thermal activation, an experiment was conducted using hot roll lamination on 8 mil aluminum lap shear samples. The rate of the hot roll and the pressure were held constant at 12 in./min (0.30 m/min) and 110 psi, respectively, and the temperature of roll was changed. After the samples were transposed via the hot roll lamination step, lap shear peak load was measured to quantify the level transposition. This lap shear data can be found in Table 5.
The data reported is just one example of conditions in which the transposable adhesive was successfully transposed. The level of cure of the activatable epoxy polymerization will be dependent on the substrate and its thickness, and the pressure, speed, and temperature of the hot roll lamination steps. The exact limits of the cure would have to be experimentally found for any given application. For this given construction and hot roll laminator settings, a temperature of 350°F was sufficient to fully transpose the adhesive. The fact that 400°F yielded the same tensile strength as 350°F suggests that the maximum tensile strength (100% cure of epoxy) is roughly 252 lbf.
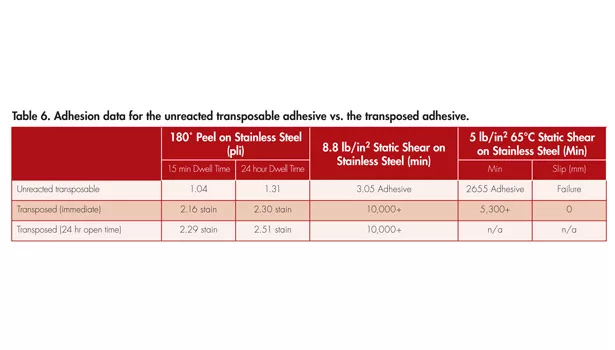
The final example to be discussed is an adhesive that transposes from a removable adhesive to a general-purpose PSA using the same base polymer as the previous two examples, and differs only by the amount of epoxy added to the system. The adhesion data is reported in Table 6.
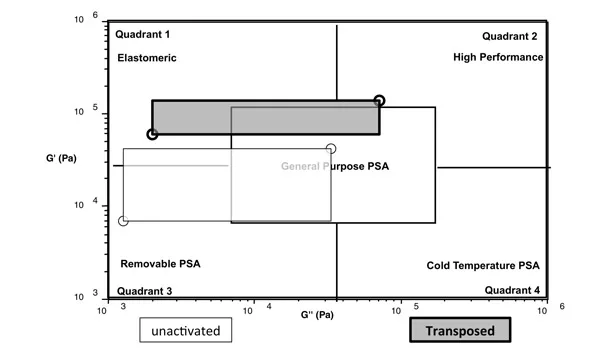
Comparable to Example 2, the peel adhesion doubles and the cohesion strength dramatically increases, supported by the room temperature and 65°C shear. Also observed is the enhanced performance independent of the open time, as expected from the results of the previous example. Revisiting the viscoelastic windows mentioned as the qualification of the types of adhesives before and after transposition, Example 3 was examined by DMA. A frequency sweep was conducted in which the performance window was constructed for the unreacted PSA and the transposed PSA. The resulting viscoelastic windows were found and displayed in Figure 3.
As seen in Figure 3, the performance window was shifted from the removable quadrant to the general-purpose region of the overall viscoelastic window plot. Thus, the transposition from one PSA state to another was evident by adhesion performance and analytical techniques.
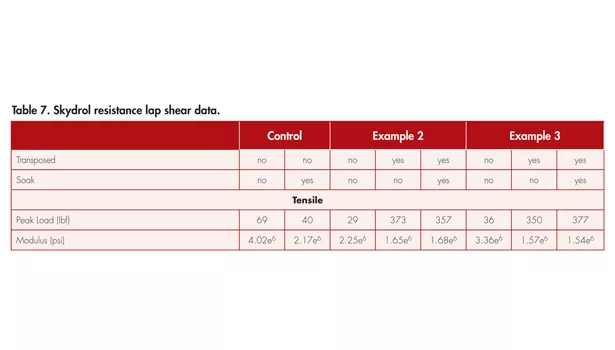
Higher temperature resistance was included in the adhesion data, but chemical resistance is another property that can also be improved by transposition. To expand on this concept, resistance to Skydrol™ was evaluated. Skydrol is an aviation hydraulic fluid consisting of phosphate esters that is known to degrade polyacrylate elastomers and PSAs.11 The need for a PSA that resists degradation by this fluid has lately been an area of focus for the tapes/adhesive industry. The 8 mil aluminum 1 x 1-in. lap shears were again used to qualify the transposable adhesive in Examples 2 and 3 before and after being soaked in Skydrol™ at 65°C for 16 hours and are shown in Table 7. The tensile data reported in Table 7 provides two key pieces of information:
• The % increase in peak load and modulus for the two examples when being transposed
• The % decrease in peak load and modulus for the adhesives after the Skydrol soak
These numbers are shown in Table 8. The degradation of the transposed adhesives was significantly better than the base adhesive, which nearly degraded to 50% of its original strength. In comparison, the transposed adhesives degraded < 5% if they did not increase in strength. (Note: a negative number for the decrease in peak load or modulus designates that the values increased.) For the cases in which there was an increase in value, we conclude that the percent increase is within the error of the test.
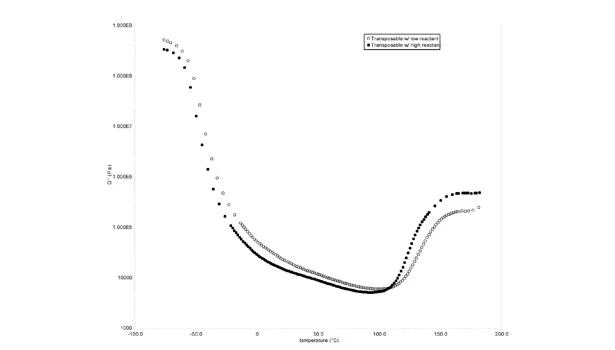
It was observed that Example 3, which had higher epoxy loading, had a lower increase in strength, which is opposite to expectations. To confirm this response to epoxy loading, the storage modulus was measured by a DMA temperature sweep of both examples before transposition. It was expected that higher epoxy loading should result in a lower storage modulus before transposition and a higher storage modulus after transposition. Figure 4 shows the resulting storage modulus curves for the temperature sweeps and verifies this prediction.
The curve that is filled in represented the example with higher loading of epoxy. As expected, the storage modulus is lower compared to its lower epoxy-loaded example before transposition and higher strength after transposition. This data confirms what was expected, but contradicts the lap shear data in Table 7. The contradiction in the lap shear data was attributed to errors in the lap shear test. Potential causes are lack of adhesion over the entire 1 x 1-in. surface due to flexibility of the 8 mil aluminum and possible volume contraction during transposition, leading to lack of adhesion over the entire test area resulting from the lack of flexibility of the aluminum.
Conclusion
Transposable adhesives were characterized physically and analytically by proving a shift of a base adhesive’s performance window as a function of storage and loss moduli. The benefit of the shift in performance window was found to be a two- to three-fold increase in peel strength in addition to dramatic increases in cohesive strength after application. The resulting transposed adhesive exhibited enhanced temperature and chemical resistance vs. the non-transposed counterpart.
It was demonstrated that the transposition process is activated by precise temperatures or UV exposure to yield a tape construction that can be manufactured and applied with long open times before being transposed to a high-strength or structural adhesive. In tape applications, this is advantageous over traditional two-part structural adhesives due to the lack of applicator guns and controlled bond lines.
For additional information, visit www.averydennison.com.
References
1. “Transpose,” Merriam-Webster.com, 2011, www.merriam-webster.com (February 28, 2013).
2. Kamperman, Marleen, Synytska, Alla, J. Mater. Chem., 2012, Vol. 22, pp. 19390-19401.
3. Burch, Robert Ray, Temperature Switchable Adhesives Comprising a Crystallizable Oil, U.S. Patent 20110265949, November 3, 2011.
4. Tunius, Mats, Switchable Adhesives, U.S. Patent 2013017246A1, January 7, 2013.
5. Therriault, Donald J., Workinger, Jane E., Water-Inactivatable Pressure Sensitive Adhesive, U.S. Patent 5,032,637, March 14, 1990.
6. Webster, Iian, Adhesives, U.S. Patent 6184264B1, February 6, 2001.
7. Chang, Pi, ACS J. Adhesion, 1991, Vol. 34, pp. 189-200.
8. Dahlquist, Carl, Proc. Nottingham Conf. on Adhesion (Maclaren & Sons Ltd., London, 1966), Part III, Chapter 5, p. 134.
9. Zajaczkowski, Michael, Lester, Christopher, Pressure Sensitive Adhesives, U.S. Patent Application 20100120931, May 13, 2010.
10. Czech, Dr. Zbigniew, Kowalczyk, Agnieszka, Munich Adhesives and Finishing Symposium 37, October 24, 2012.
11. Solutia Inc., Skydrol, www.skydrol.com/pages/materials_chart.asp (February 28, 2013).
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!








