Improved Plasticizers Help to Fill the Gap in Polysulfide Sealants

Polysulfide sealants are known for their excellent resistance to solvents, chemicals, low temperatures, and flex-cracking, as well as good adhesion, elasticity, and impermeability. These characteristics make them a valuable solution in applications such as insulated glass seals, construction joints and sealants, aerospace (particularly sealants for aircraft fuel tanks and bodies), and secondary containment.
Polysulfide sealants for insulated glass and construction are primarily offered in a two-part package. Both parts contain plasticizer to deliver essential performance characteristics to the sealant.
- In the “A” (polymer) side, the plasticizer functions to soften the polymer, control the viscosity, and allow higher filler loading for economy.
- In the “B” (catalyst) side, the plasticizer serves as a carrier for the catalyst used to cure the sealant, ensuring good mixing, viscosity control, and package stability.
While the role of the plasticizer is critical, safety concerns have plagued the plasticizer chemistries that have historically been favored for polysulfides. These include benzyl phthalates and certain chlorinated paraffins, which are increasingly subjected to regulatory pressure and restrictions.
Recently, novel monomeric and oligomeric dibenzoate plasticizers have been introduced; these new plasticizer solutions have been specially tailored for polysulfide sealant applications. These plasticizers have been found to be high-performing, low-fogging alternatives to traditional chemistries used in polysulfides. Additionally, they are expected to be more environmentally friendly, safer, and REACh-compliant alternatives to chemistries such as chlorinated paraffins. Data is presented below to demonstrate the performance of one of these recently commercialized plasticizers.
Evaluating Performance: Novel Oligomeric Dibenzoate Plasticizers in Polysulfide Sealants
Commercially available as Kalama® VITROFLEX® benzoate plasticizers
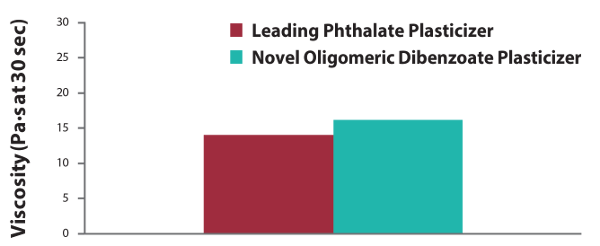
Figure 1. Viscosity characteristics equivalent to phthalates in the B-side of polysulfide sealant.
Both components of a polysulfide system, the polymer side and the catalyst side, must have similar viscosities for even mixing. B-side polysulfide sealant formulations formulated with the new oligomeric benzoate plasticizer exhibit similar viscosity characteristics to the leading phthalate plasticizer. In addition, the formulations exhibit excellent viscosity stability in centrifuge tests (1,000 rpm for 60 minutes), with no signs of settling.
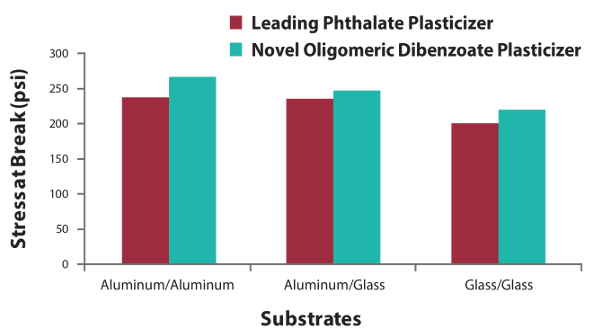
Figure 2. Increased lap shear strength in the B-side of polysulfide sealant.
Replacing a phthalate plasticizer with the new oligomeric dibenzoate plasticizer in the B-side resulted in cohesive failure and improved lap shear stress at break, demonstrating good adhesion to substrates such as aluminum and glass.
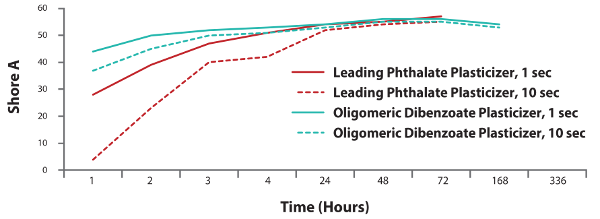
Figure 3. Faster hardness development in the B-side of polysulfide sealant.
The oligomeric dibenzoate plasticizer provides rapid cure rates and accelerated cure-through in polysulfide formulations.
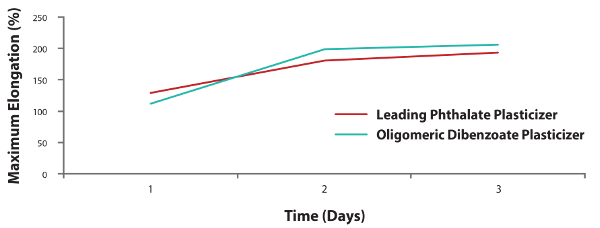
Figure 4. Excellent flexibility (ASTM D412 Elongation Method) in the B-side of polysulfide sealant.
Polysulfide formulations with the oligomeric dibenzoate plasticizer maintain excellent adhesion while providing flexibility for tailored performance in insulated window sealant applications.
Conclusions
Compared to a leading phthalate plasticizer, the newly developed oligomeric dibenzoate plasticizer exhibited equivalent or enhanced performance for characteristics that are highly desirable in polysulfide sealants: viscosity, adhesion, fast cure, and elasticity. In addition, two recently launched plasticizers for the A-side are now available. These commercially available A-side plasticizers have been specially tailored for high permanence, low migration, and low odor.
Together, these new plasticizers offer a complete package option to optimize performance in both components of polysulfide sealants. In addition to performance and economy, these plasticizers provide formulators with the option to replace chemistries under pressure from regulatory restriction and consumer preferences.
Contact the author:
Emerald Kalama Chemical
kflex@emeraldmaterials.com
+1 360-954-7100
www.emeraldkalama.com
Disclaimer: The information contained herein is believed to be reliable, however is based upon laboratory work with small scale equipment and does not necessarily indicate end-product performance. Because of variations in methods, conditions and equipment used commercially in processing these materials, Emerald makes no representations, warranties or guarantees, express or implied, as to the suitability of the products for particular applications, including those disclosed, or the results to be obtained. Full-scale testing and end-product performance are the responsibility of the user. Emerald Performance Materials shall not be liable for and the customer assumes all risk and liability for use and handling of any materials beyond Emerald’s direct control. Nothing contained herein is to be considered as permission, recommendation nor as inducement to practice any patented invention without permission of the patent owner.
Test Methods
|
A Side Preparation |
Mix polymer and liquid components on low speed until well mixed. Add filler in portions, making sure each portion is well blended before adding the next. Continue mixing for 20-25 minutes or until the Hegman fineness is at least 4. |
|
B Side Preparation |
Blend raw materials for 10 minutes at 2,000 rpm using a dispersion blade. Grind the dispersed mixture on a three-roll mill until the Hegman fineness is > 4. |
|
Sealant Preparation |
Mix the A and B side together in a 10:1 volumetric ratio until the mixture is uniform in color. |
|
Pot Life (Mixing Method) |
A 10:1 volumetric ratio of A to B side was mixed together until the color was uniform. The sample was mixed periodically until the mixture hardened or became sticky to the point where it was no longer spreadable. This time was recorded as the pot life. |
|
Pot Life (Steady Shear to 250 Pa·s) |
The time was recorded for a 10:1 volumetric ratio of A to B side to have uniform color. Then the mixture was immediately placed onto a rheometer using parallel plate geometry. The time at which the viscosity was 250 Pa·s was recorded and added to the initial mixing time. The parallel plates were set at a gap of 100 μm and run at a steady shear of 100 s-1. |
|
Shore A Hardness |
A 10:1 volumetric ratio of A to B side was mixed together until the color was uniform, then passed through the three-roll mill once. The sample was then spread evenly into an aluminum pan, covered with a polyethylene sheet, turned upside down, and pressed on a glass plate. 1s and 10s readings were performed every hour between 1-4 hours, then every day for seven days, followed by a final 14-day measurement. |
|
Tensile Strength |
A 10:1 volumetric ratio of A to B side was mixed together until the color was uniform, then passed through the three-roll mill once. The sample was then pressed between two glass plates lined with a polyethylene sheets and 3.8 mm shims. After curing 24 hours, specimens were prepared using the ASTM D412-C die and pulled on the tensometer at 20 in./min. |
|
Lap Shear |
A 10:1 volumetric ratio of A to B side was mixed together until the color was uniform, then passed through the three roll mill once. The sample was applied to a 1 in2 area of substrate. Then, a second substrate was applied to the top and placed in a jig following ASTM C961. Six specimens were cured at room temperature for two days, then in an oven set at 70°C for six hours, and cooled overnight at ASTM standard conditions. Specimens were pulled to failure on a tensometer set at 0.5 in./min. |
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!


