Going Green

Pine chemicals are renewable, naturally occurring materials derived from the pine tree (genus Pinus). The range of chemical classes obtained from pine trees includes numerous plant sterols, terpenes (or turpentine), resin acids (or rosin) and fatty acids. Pine trees originated in the Northern Hemisphere, but are now found worldwide. Significant pine resources exist in the United States, Canada, South America, Scandinavia, parts of Spain and throughout Asia. All these regions have pine chemical production capabilities, though China continues to be the world's largest national producer of pine chemical raw materials.
The earliest recorded use of pine pitch was in the 4th through 2nd centuries B.C. in Greece, Asia Minor and Egypt. Pine resins were used in the 4th century A.D. as adhesives for attaching colored stones to buildings.1 As the number of wooden ships in the European navies grew, so did the need for rosin, pine tar and pitch. These products, which were essential for waterproofing and caulking the vessels, became known collectively as naval stores, a term still used today. The high-quality pine chemicals available today were initially developed and produced in the mid-to-late 20th century.
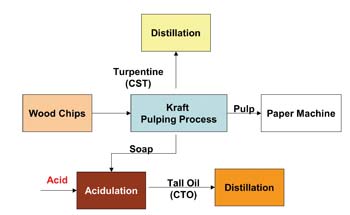
COMMERCIAL SOURCING OF PINE CHEMICALS
Rosin and turpentine are obtained by three means: by tapping live trees (scoring a portion of the trunk and collecting oleoresin), by isolating co-products of the process used to manufacture Kraft paper (see Figure 1), and by extracting pitch-containing pine stumps. Various chemical and manufacturing processes, including acidification, fractional distillation, and solvent extraction, are used to obtain the purified forms of the different chemicals.The process to isolate the paper-making co-products crude tall oil (CTO) and crude sulfate turpentine (CST) is the most efficient and has the best long-term outlook. CTO is a mixture of rosin, fatty acids and pitch. Interest in pitch has increased following the discovery that it is an economically viable source for plant sterols, an active ingredient in functional foods and an intermediate in steroid pharmaceuticals.
Resins and rosin acids, plant sterols, and saturated fatty acids are generally solids; unsaturated fatty acids and terpenes are liquids (see intro page photo for examples of solid pine chemical derivatives for adhesive applications). Pine resins for industrial use can be packaged in forms ranging from molten, bulk trucks and railcars to 50-lb paper bags. Pine chemical raw materials are infinitely renewable as long as they are sourced from properly managed, sustainable forests like those in many parts of North America and Europe.

MARKET OVERVIEW
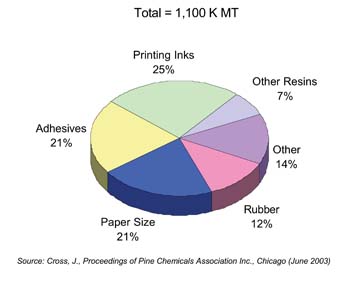
The annual global production of major pine chemical raw materials, crude tall oil (CTO), crude sulfate turpentine (CST) and crude gum rosin is estimated to be 2,600 K MT.3 Of this 1,100 K MT is converted to rosin, 500 K MT is converted to fatty acid, and the balance is converted to a range of terpenes, oils, flavor and fragrance chemicals, and bio-fuels.
Rosin and its derivatives account for the largest segment of the pine-chemicals market. Major uses include adhesives, printing inks, coatings, surfactants, rubber, paper products and chewing gum (see Figure 2). Within adhesives, the packaging and bookbinding segments (see Photos 1 and 2) account for most consumption, followed by pressure-sensitive and construction adhesives. Uses for terpenes and derivatives include adhesives, flavors, perfumes, camphor and cleaning products; tall oil fatty acid (TOFA) and derivatives are used to make polyamide resins, fuel additives, lubricants, and alkyd paints.


PINE CHEMICALS IN THE ADHESIVES MARKET
There are three major classes of tackifier resins for the adhesives industry: terpene, hydrocarbon and rosin resins. Terpene (pine-based) and hydrocarbon (petrochemical-based) resins are both hydrocarbons; that is, they contain only carbon and hydrogen. While they are similar in that respect, they impart somewhat different properties to the resultant adhesives. Terpene-based resins are more diverse than petrochemical hydrocarbons in that they are readily modified with other chemicals (e.g., phenol) to produce an array of products. Rosin resins significantly differ from the previous two types in that they contain carboxylic acid and/or ester groups. These resins are generally more polar and narrower in molecular weight, making them excellent tackifiers for a variety of end-use applications.
All groups of pine chemicals, except plant sterols, are used by the adhesives and sealants industry. Rosin resins, including esters and polymerized rosins, are used as tackifiers to modify the properties of selected polymers to produce adhesives and sealants. Polyterpene resins are used to modify non-polar polymers for these same applications. Tall oil fatty acids can be dimerized to produce dimer fatty acids that, in turn, are the major ingredient in thermoplastic polyamide adhesives.
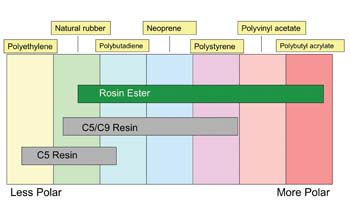
End users expect hot-melt packaging adhesives to run faster and perform on a range of substrates. Terpene phenolic resins, derivatives of alpha-pinene, can deliver improved adhesion to difficult substrates such as recycled cardboard. They offer better green strength, making them useful for high-speed packaging lines with short set times (see Figure 3). Rosin esters are commonly used to increase adhesion and the temperature performance range of ethylene vinyl acetate (EVA)-based adhesives. Rosin esters have the added benefit of being compatible with a range of polymers, thus limiting formulating complexity
(see Figure 4).
In pressure-sensitive adhesives, the tight supply of styrene-isoprene-styrene (SIS) polymer has many formulators considering alternative systems like styrene-butadiene-styrene (SBS) and styrene-isoprene-butadiene-styrene (SIBS). This trend presents opportunities for pine chemicals like rosin ester and polyterpene resin because they have good compatibility and performance in these alternate polymer systems.
The compatibility of rosin resins with polymers used in aqueous adhesives makes them well suited for these new systems. As new or modified polymers continue to be introduced, new waterborne resins will be developed to meet the challenge. Resins for waterborne adhesive can be purchased as dispersions or neat. Some pine chemicals are used in radiation-cured systems, but this remains a relatively small part of overall demand. Pine chemicals, like rosin ester and terpene phenolic resins, have been used in radiation-cured acrylic adhesives.

ABOUT ARIZONA CHEMICAL
Arizona Chemical is the world's largest producer of pine chemical raw materials and derivatives. In 1930, International Paper and American Cyanamid formed Arizona Chemical to mine salt cake in Camp Verde, AZ (see Photo 3). By 1936, after the mine was closed, Arizona Chemical began processing papermaking co-products. Arizona Chemical's aggressive global growth strategy began in 1985; since then, the company has significantly increased its number of manufacturing locations through acquisitions in the United States and Europe.
Today, the company's global and European headquarters are in Jacksonville, FL, and Almere, The Netherlands, respectively. The company has 12 production facilities (six in the United States and six in Europe); two laboratories (one in the United States and one in Europe); and sales offices around the world. Arizona Chemical produces and globally distributes a range of products for many of the applications discussed in this article
For more information on pine chemicals, visit the Pine Chemicals Association Inc.'s website at
http://www.pinechemicals.org. For more information on Arizona Chemical, visit http://www.arizonachemical.com or e-mail azcadhesives@ipaper.com.
REFERENCES
1 Zinkel, D.F.; Russell, J.R., Eds., Naval Stores, Production, Chemistry, Utilization, Pulp Chemicals Assoc., NY, pp. 3-29 (1989).2 Turner, J.M. Proceedings of 2003 Pine Chemicals Association, Chicago, Pine Chemicals Association Inc., section 4 (2003).
3 Arizona Chemical Internal Estimates
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!



