Evaluating Compatibility of UV-Cure PSAs With Silicone-Release Technologies


The sudden accelerated emergence of UV-PSAs is a result of three historical factors that are all coming together today:
1. The technical development of new chemistries by the raw material suppliers for the UV-PSA industry.
2. The coordinated commitment by the larger adhesive manufacturers for the necessary time, funding and people resources for developing quality UV-PSAs.
3. Increasing regulatory pressures from the local, state and federal environmental-protection programs.
The beneficial result of these events is that there are now for the first time high-performance UV-PSA alternatives that provide complete air-pollution control, and deliver measurable productivity gains with reduced need of manufacturing space — and all this with improved PSA performance benefits. Most exciting are the unique possibilities for tandem-coating processes with UV/EB-curable release coatings, UV-PSAs and UV-cure printing inks. It is now technically possible in one continuous UV or EB procedure to prepare printed label stock laminations at line speeds well over 1,000 fpm without the presence of even one process oven!
Attempts at developing a full-range of UV-PSAs encompass three categories of application methods and final performance:
Liquid UV-PSAs:
These are room-temperature-coatable syrups with viscosities of about 1,000-10,000 cps. UV-curing crosslinks these tackified monomer/oligomer compositions into durable PSAs with a desirable balance of tack, peel and shear adhesive properties.Warm-Melt UV-PSAs (UV-WMPSAs):
These are essentially modified hot melt PSAs that liquefy at relatively low application temperatures (~200°F-250°F). They regain their high-performance adhesive properties upon UV-induced crosslinking for reinforcing the elastomeric phase of the PSA matrix. The key benefits from this technology are faster processing/higher productivity, and the ability to coat thermally sensitive liner and face stock films.Hot Melt UV-PSAs (UV-HMPSAs):
These are similar to typical hot melt PSAs with their standard application temperatures (~300ºF-350°F). However, conventional HMPSAs inherently have poor resistance to solvents, plasticizers and heat. The major benefits from post-application UV-exposure are to improve the adhesive’s high-temperature shear and working-life performance along with improved chemical resistance by reinforcing the polymer networks with chemical crosslinks. Superior performance at elevated temperatures (>300°F) is allowing UV-HMPSAs to be used for the first time in high-temperature tape applications (e.g., automotive masking tape).Early attempts at formulating UV-PSAs did not succeed in replacing standard high-performance PSAs, but they were able to at least provide performances sufficient for the requirements of many of the less demanding applications (such as screen-printable PSAs). The major difficulty was the development of high-tack formulations combined with a desirable balance of shear-strength and peel-adhesion properties on par with standard-chemistry “permanent” PSAs. Because of this, the liquid UV-PSA market potentials have remained limited and they will remain so until the technology is developed to produce UV-PSAs that perform at least as well as the standard, non-UV PSAs.
The first liquid UV-PSAs had a number of undesirable features that made their acceptance by the adhesive market difficult. They simply did not meet the broad performance criteria required by many adhesive tape-and-label applications as supplied by most conventional PSAs. •
- They had very high viscosities (>>20,000 cps). •
- They displayed sub-par adhesive properties. •
- They were strongly inhibited by oxygen. •
- UV exposures often resulted in only partial cures, especially for thicker PSA applications. •
- They maintained poor catalyzed bath lives. •
- Their tack-adhesion characteristics were often achieved by under-curing, resulting in poor cohesive strength and eventual loss of tack over time.
The earliest UV-WMPSAs and UV-HMPSAs were often thermally unstable and would readily gel in the melting tank. Their overall adhesive-performance profiles and their processability were less than impressive. However, innovative polymer and chemical developments over the past 10 years have brought about the emergence of new PSA product offerings with new and improved performance: •
- Thermally stable photoinitiators; •
- Stabilizing additives (e.g., antioxidants and inhibitors); and •
- Better adhesive-polymer designs for providing balanced fast-cure response with functionally desirable PSA properties.
The challenge for all formulators of UV-PSAs is to find a way to induce polymerization to the necessary high molecular weight for exhibiting the tough viscoelastic behavior required by high-performance PSAs. The trick is to do this without the technique of introducing high multifunctional crosslinking that results in too high of a glass transition temperature (Tg) with poor elastic modulus (i.e., too rigid).
Testing and Results Procedures
We now understand the historical background and processing impact that emerging UV-PSA technologies have in store for the converter. However, there is a very simple but crucially important question that remains to be answered: How do these new UV-PSAs perform against silicone-release-coated liners for use with labels and tapes? (See Figures 1 and 2.)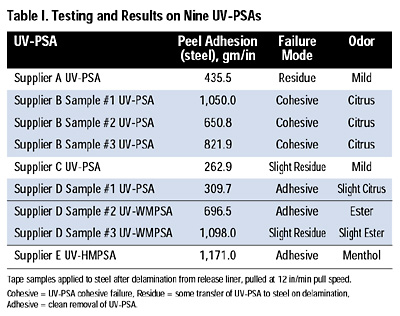
At the request of a number of end users of our various release-coating chemistries, we initiated a program to evaluate the release performance of today’s emerging, developmental UV-PSA products against representative silicone-release coating systems available from Rhodia Inc. A total of nine free-radical-curing UV-PSAs from five different companies were obtained. These nine UV-PSAs can be classed according to three categories of process and performance: liquid UV-PSAs, warm melt UV-WMPSAs (~200°F) and hot melt UV-HMPSAs (~300°F+). See Table I.
In order to best differentiate the release performances of these UV-PSAs, we chose five sets of silicone-release-chemistry and liner combinations: •
- UV epoxy-silicone “premium” release, •
- UV acrylate-silicone release, •
- PC-267 thermal solventless release, •
- PC-107 silicone-emulsion release, •
- Three families of ‘differentially’ tight release.
All UV-PSAs were direct-cast UV-cured onto the five categories of silicone-release liners after laminating with 1-mil PET film in the roller nip on our Euclid coater with a nip pressure of 160 psi and the rollers turning at their slowest speed. These laminates were then cured with two Fusion H-bulbs (300 watts/in) @ 20 fpm. In the case of the UV-WMPSAs and the UV-HMPSAs, samples were reheated in our oven followed by a second pass through the pressurized Euclid nip. The liquid UV-PSAs were prepared with final thicknesses of about 1-2 mils. The UV-WMPSAs and UV-HMPSAs were prepared with final thickness of about 2-4 mils. Full cure was realized for all these UV-PSAs by determining that a second pass of UV-curing produced no noticeable change in the performance of the UV-PSA.
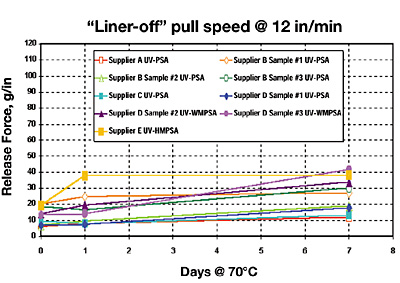
Results I: UV Epoxy-Silicone “Premium” Low Release on 2-mil PET Film
This first section looks at the initial and aged release performance of these free-radical UV-PSAs against our recommended “premium” low-release UV/EB cationic epoxy-silicone chemistry (Figure 3).The UV epoxy-silicone “premium” releases on polyester film show remarkably stable aged release for all nine UV-PSAs at 12 in/min pull speed. Even after seven-days aging @ 70°C, the highest measured release for any UV-PSA is under 45 gm/in release force. Although these UV-PSAs were direct-cast coated and UV-cured directly onto the UV-silicone-release coating, there appears to be no chemical reaction between the two different UV-curable chemistries (cationic vs. free-radical).
The WMPSAs and the HMPSAs give a slightly higher release force with the UV-silicone-release-coated polyester-film liner. This can be attributed to the fact that the WMPSAs and HMPSAs were direct-cast applied onto the silicone liner at heavier coat weights than were the liquid UV-PSAs (our method of application could not achieve lower coat-weight lay-downs for these high-viscosity warm melts and hot melts). This made them softer and more elastic than the liquid UV-PSAs. This effect is more physical in its aged-release nature and not due to chemical silicone-PSA release instability at the interface (note that there was no major increase in accelerated-aged release from one day to seven days @ 70°C). I would expect that if we had the proper warm melt/hot melt coating equipment for laying down a lower coat weight, this phenomenon would either be reduced or eliminated.
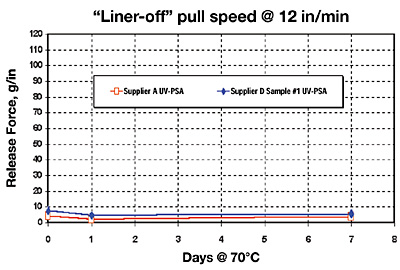
Results II: UV-Acrylate Silicone-Release on 42# Rhi-Liner SCK Paper
This section looks at the initial and aged release of two UV-PSAs against our new UV/EB free-radical acrylate-silicone low-release chemistry (Figure 4, page 32).The first thing to be aware of is that the level of achieved release is extremely low (less than 10 gm/in), and no build-up phenomenon whatsoever has been observed under our testing conditions with both evaluated UV-PSAs. This is both surprisingly unexpected and of beneficial importance. Our initial expectations were that a free-radical-curing UV-PSA would naturally react to the surface of a free-radical-curing UV-silicone-release coating – this should result in liner “lock up” whereby the adhesive would fail to release against the UV-silicone layer.
These results, however, indicate that the UV acrylate-silicone chemistry is reacting to a very complete and stable level such that there is no residual acrylate functionality on the silicone polymer available to react with the direct-cast and cured UV-PSA. Or, if there is any residual acrylate functionality left on the silicone polymer, it remains well shielded within the crosslinked matrix of the release coating under methyl-rich segments of the silicone polymer that are efficiently orienting themselves to the surface. In either case, the UV-PSA chemistry does not appear to be chemically reacting with the UV acrylate-silicone.
Results III: Thermal Solventless Silicone-Release
This section looks at the initial and aged release of the UV-PSAs against our PC-267 thermal solventless release (platinum-catalyzed hydrosilation, vinyl and hydride-functional silicones) on 42# Rhi-Liner SCK paper (Figures 5 and 6).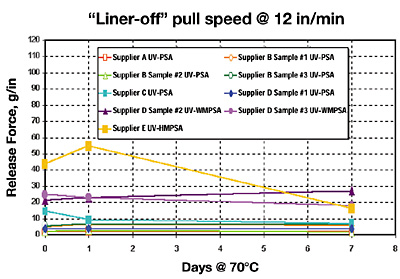
Adhesive coat weight has some influence on the net release level. The three adhesives that were similar to a true hot melt adhesive gave a higher release level. This is due in part to our inability to apply a uniform adhesive coat weight. In short, thermal, solventless release coatings should be very compatible and predictable with the new pressure sensitive UV-cured liquid and hot melt adhesives.
Results IV: Silicone Emulsion Release
This section looks at the initial and aged release of the UV-PSAs against our PC-107 silicone-emulsion release chemistry (Figures 7 and 8, page 35).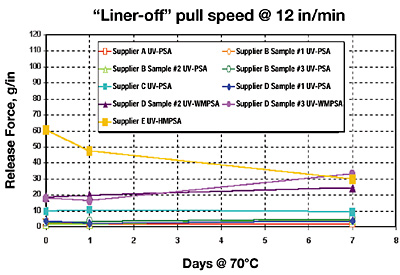
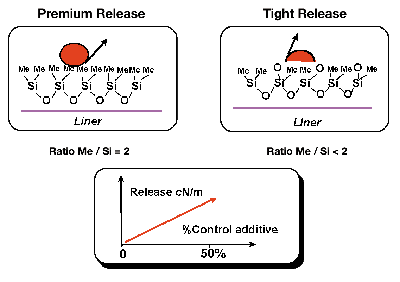
Results V: “Differential” Tight Release
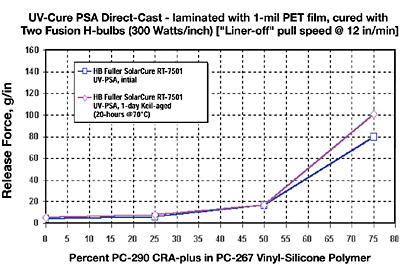
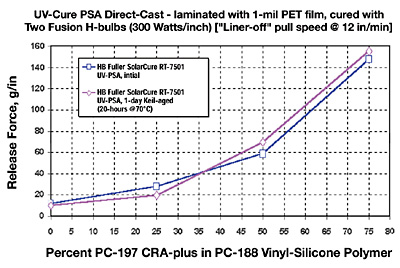
This section addresses particularly the question as to whether silicone-release coatings for UV-PSAs can be modified for three chemical families with incrementally tighter differential releases. The approach was to prepare a series of four differential levels of release ranging from “premium” low to sequentially tighter levels of release. This is accomplished by the blending of CRA-plus (control release additive “plus”) additives to the three different base “easy release” silicone polymers (Figure 9).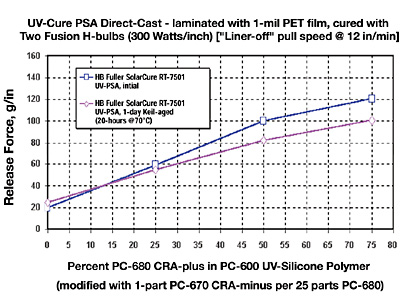
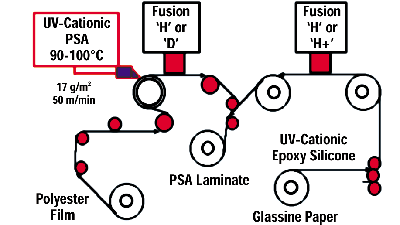
Results VI: UV-Cationic PSAs with UV-Cationic Silicone-Release Coatings
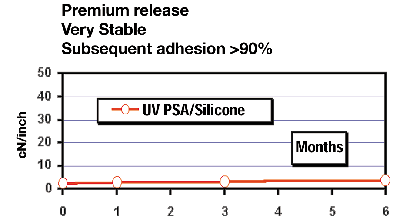
Our efforts in France have been focused on the release interactions of UV-WMPSAs based on our iodonium-borate cationic photoinitiator [Rhodorsil 2074] with our UV-silicone cationic release coatings based on the same photoinitiator. The UV-WMPSA was based on epoxy-functional Kraton™ polymers synthesized by Shell Chemical Co., Houston, as these are tackified with a tackifying resin of a softening point of 97ºC-103°C.Approximately 1-mil of this adhesive was applied at 100°C at a viscosity of about 1,000 cps using a Meltex five-hole “buse” applicator onto polyester film. This UV-cure tape was then laminated “in line” with our cationic UV epoxy-silicone-release-coated glassine paper liner (Figure 13).
Conclusion
Interest is growing in environmentally friendly UV-curable pressure sensitive adhesives. Conference papers on the development and application of UV-PSAs are becoming a very serious technical focus. Adhesive manufacturers and their raw material suppliers are committing concerted efforts and resources towards the new-product development of high-performance UV-PSAs. Customer inquiries on the performance of silicone-release liners against UV-PSAs are increasing.Many of the UV-PSAs we evaluated can for the most part be considered very good as to their general tack- and peel-adhesive performance. But some of them were particularly soft (especially the liquid UV-PSAs) such that removal of their tapes from a polished-steel plate left a good deal of undesirable residue. Those that were tougher in constitution and that did not leave a lot of residue upon removal were generally of lower peel-adhesion strength. Almost all of them had characteristic odors ranging from mild to strong. This may be indicative of either unreacted UV-monomers or to the post-UV by-products of the photoinitiator.
We have found that many of these emerging UV-PSAs perform very well against four different chemical families of Rhodia’s silicone-release-coated liners – UV cationic epoxy-silicones, UV free-radical acrylate-silicones, thermal solventless silicones and thermal emulsion silicones. The very beneficial stable releases achieved from direct-cast applied and cured UV-PSAs promise many potential processing advantages to be had with different combinations of these two converting technologies (such as in-line tandem coating). By providing a systems understanding of the performance of Rhodia’s silicone-release technologies with these emerging UV-PSAs, we mean to deliver a valuable resource to our customers.
We encourage the introduction of UV-PSA technology to be as robust and as “systems compatible” as possible. It is one thing to just simply introduce quality UV-PSAs to the converting market. It is preferable to introduce quality UV-PSAs that are compatible with a converter’s existing process equipment, liners and face stocks, and wide-selection of silicone-release technologies.
Acknowledgements
The authors wish to recognize the extensive contributions to the global release-coatings application team within the Rhodia organization, especially to such dedicated laboratory and pilot-coater technicians as Butch Burton, Jamie Ellison and Dwight Payseur. We also express our gratitude to those UV-PSA manufacturers who submitted their developmental and commercial UV-PSAs for our evaluation.This article is based on a presentation made at RadTech 2000, April 9-12, 2000, Baltimore, Md.
Additional information on silicone release coatings is available from Rhodia Inc., 911 E. White St., PO Box 11674, Rock Hill, SC 29731; phone 800-476-5260, ext. 238; fax 803-329-5269; e-mail rgriffit@us.rhodia.com; Web site www.silicones.us.rhodia.com.
Links
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!



