THE JOINING OF MEDICAL PLASTICS
When Alexander Parkes developed the first man-made plastic in the 1860s, he had no idea what an integral role the material would come to play in daily life. Plastics today play an important part in cutting-edge technologies such as the space program, bullet-proof vests and prosthetic limbs, as well as in everyday products like beverage bottles, automobiles, and particularly medical devices.
Plastics use in medical devices has sustained an average annual growth rate far in excess of the Gross Domestic Product (GDP). U.S. demand for medical plastics will increase nearly 3% per year to 3.6 billion pounds in 2008, valued at $5.0 billion.
Key drivers of medical plastics usage include the following.
- Aging of the U.S. population
- Continuing cost reduction pressures in the healthcare field
- Advances in polymer performance
- Introduction of new and often life-saving devices
- Environmental/disposable/non-disposable medical device triad
Hard-to-bond plastics like polyolefins are most often assembled using adhesives. While adhesives are the most versatile assembly method for plastics, only a few industrial adhesives offer suitable bond strengths on hard-to-bond plastics. Cyanoacrylate, light-curing cyanoacrylate, hot-melt and light-curing acrylic adhesives have typically been used with typical difficult-to-bond plastics.
To make an adhesive formulation, the fluid adhesive should have a surface tension no higher (and preferably 10 mN/m lower) than the critical surface tension of the solid adherend.
Adhesion impediments with medical plastics include:
- Low surface tension
- Migration of formulation components
- Mold release agents
- Low temperaturev
- UV light
- Mechanical stresses
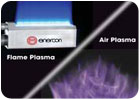
Flame plasma systems are comprised of a combustion/electrical station and a burner assembly, manufactured with two primary burner configurations: ribbon and enhanced velocity. A flame plasma is formed when a flammable gas and atmospheric air are combined and combusted to form an intense blue flame. Medical plastics are made polar as species in the flame plasma affect electron distribution and density on the surface. Polar functional groups such as ether, ester, carbonyl, carboxyl and hydroxyl are contained in a flame plasma; these are incorporated into the surface and affect the electron density of the polymer material. This polarization and functionalization is made through reactive oxidation of a surface.
Atmospheric chemistry plasma systems generate a chemical plasma field in an electrically charged atmosphere. This method blends gases that deposit various chemical groups on the substrate surface to improve its surface energy. Chemical plasma treatment is essentially a nano-cleaning, micro-etching and functionalizing process that provides different surface characteristics depending on the gas chemistry employed.
For more information on surface treatment, contact Enercon Industries at (262) 255-6070, e-mail info@enerconind.com, or visit www.enerconind.com.
Polymers: gc (20°C): mN/m:
Polymers:Poly(tetrafluoroethylene) gc (20°C): mN/m:18
Polymers:Silicone, polydimethyl gc (20°C): mN/m:24
Polymers:Poly(ethylene) gc (20°C): mN/m:31
Polymers:cis-Poly(isoprene) gc (20°C): mN/m:31
Polymers:Poly(styrene) gc (20°C): mN/m:33
Polymers:Poly(vinyl alcohol) gc (20°C): mN/m:37
Polymers:Poly(methyl methacrylate) gc (20°C): mN/m:39
Polymers:Poly(vinyl chloride) gc (20°C): mN/m:40
Polymers:Poly(acrylonitrile) gc (20°C): mN/m:44
Polymers:Amine-cured epoxide gc (20°C): mN/m:44
Polymers:Poly(ethylene terephthalate) gc (20°C): mN/m:45
Polymers:Cellulose gc (20°C): mN/m:45
Polymers:Poly(hexamethylene adipamide)
Polymers: (nylon 6,6) gc (20°C): mN/m:46
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!



