Ionic Times

Editor’s Note: Look for more coverage of this exciting topic in future issues of ASI.
Ionic liquids are a hot topic in scientific research. They have been discussed as revolutionary materials offering a unique set of properties. BASF’s BASIL™ technology involves ionic liquids, and solves a common problem in many production processes: the removal of acids that are formed as byproducts. The conventional method results in the formation of solid salts, which cause problems in large-scale production. With the BASIL technology, the salts remain liquid and are much easier to handle.
This article addresses several frequently asked questions about ionic liquids.

What Are Ionic Liquids?
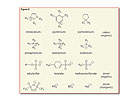
Ionic liquids are purely ionic, salt-like materials, which are, per definition, liquid below 100°C. They generally have melting points below room temperature, with some even below 0°C.Comparing a typical ionic liquid, such as 1-ethyl-3-methyl-imidazolium chloride (m.p. ~80°C) for better comparability to sodium chloride, with a typical inorganic salt, such as table salt (NaCl, m.p. 801°C), it is apparent that the imidazolium cation has an asymmetric shape and a delocalized positive charge. Consequently, solidification of the ionic liquid will take place at lower temperatures. The strong ionic interaction within ionic liquids results in a negligible vapor pressure unless decomposition occurs and makes the material non-flammable and highly stable thermally, mechanically, and electrochemically. Furthermore, it imparts desirable solvent properties and, depending on the cation and anion’s nature, can generate anything from immiscibility to water up to immiscibility of unpolar organic solvents that results in biphasic systems (see Figure 1).
How Pure Are Ionic Liquids?
What exactly is purity? Purity is usually defined by the actual content of the desired compound expressed in weight percent. This is not an easy thing to do with ionic liquids. As salts, they intrinsically consist of two compounds: a cation and an anion. For example, a sample of EMIM Cl might contain 5 wt. % of EMIM HSO4 as impurity. This means the ionic liquid is 100 wt. % pure in terms of the cation EMIM and 95 wt. % pure in terms of the anion chloride.Weight % is only one of many possible definitions of purity. A catalysis chemist would define purity as being free of any coordination species, such as halides, which deactivate the metal by formation of stable complex compounds. An electrochemist would define purity as having no oxidizable impurities, which narrow down the electrochemical window. An engineer might prefer not to have impurities that affect viscosity. An end user will define purity as being free of residual, potentially toxic alkylating agents. These examples show that only the targeted application defines what purity means. In the end, ionic liquid manufacturers do not sell purity, but performance.
Does "Technical Grade" Mean "Impure"?
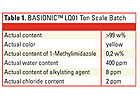
BASF, for example, provides large-scale quantities of ionic liquids at a “technical grade.” These materials have a certain quality derived from the actual manufacturing process. For example, the parameters for BASIONICTM LQ01 (EMIM EtOSO3) from a typical ton scale production are shown in Table 1.This shows that the initial quality is already very high and “technical grade” does not mean “impure.” However, as mentioned, the actual quality must meet specific customer needs. For example, actual content and color for a given application might not be relevant, but the water content must be below 50 ppm. In this case, the quality requirements are less challenging regarding actual content, but very demanding regarding water. It not only makes no sense, but it is impossible to provide a purity from the beginning that will meet all imaginable future quality demands.
Is the Color of Ionic Liquids a Problem?
Colored materials are often perceived as impure. Most ionic liquids are colorless liquids, but tend to become colored, especially during prolonged thermal treatment. However, the color persistently stays in the ionic liquid and cannot be extracted in any organic product or solvent. As of now, the colorant cannot be isolated because the quantities are just too low. It is thought that oligomers of the imidazole-or even radical ions-might cause the color. For commercial applications, the color is usually not a problem, since products are not affected.BASF has been continuously running a pilot plant for an extractive distillation process for three months. The color of the ionic liquid changed to black, but the performance stayed constant without the need for any purge. During the entire run, the product has been colorless, clear and fully in line with the required specification. For this application, it would have been a waste of money to start with a colorless high-price ionic liquid, as the colorless ionic liquid would also have turned black under the required high temperatures. In cases where color does matter for the application, BASF has developed technological know-how to produce near colorless ionic liquids on demand.
How Stable Are Ionic Liquids?
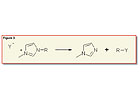
Ionic liquids generally show remarkably high thermal stabilities of >200°C. A decomposition pathway of the usually very stable imidazolium-based ionic liquid is the de-alkylation of the cation (see Figure 3).
Under basic conditions, imidazolium-based ionic liquids tend to form carbenes, which can undergo further decomposition, such as irreversible disproportionation (see Figure 4). Are ionic liquids toxic?

Are Ionic Liquids Toxic?
There is no general answer to this question. Since ionic liquids can consist of many chemically different types of cations and anions, this question must be answered case by case. The ‘magic ionic liquid’ that meets all requirements in toxicity, ecotoxicity, stability and performance simply does not exist. Until now, there is still very limited information available regarding a full toxicological profile of ionic liquids. Only recently has some data been published that was collected for the notification process of the corresponding ionic liquids and represents examples from the most common classes of cations: EMIM, BMIM and ammonium (see Table 2).These examples show that ionic liquids can be toxic as well as completely harmless and biodegradable. However, the harmless ionic liquids may not necessarily provide the best performance. In the end, a balanced decision between toxicological and performance properties has to be made to gain the best fit to the targeted application. Some ionic liquids may only be suitable for being handled by skilled personnel in chemical plants; others for a use closer to the end user will have to be non-toxic and readily biodegradable.

The
BASF Intermediates operating division has developed a process called BASILTM,
a concept to remove acids from reactant mixtures. The developers of BASIL
received the BASF Innovation Award in October 2004, and the Innovation Award
sponsored by the journal European Chemical News.
Are Ionic Liquids Green?
Besides their technical and economical potential, ionic liquids are often said to be green solvents or green materials. The “greenness” has often been justified by ionic liquids having no vapor pressure, hence they are not released into the environment by evaporation. There certainly might be applications where this property can help to make a particular process greener than a corresponding process with organic solvents. However, this is not necessarily (and not generally) the case. It might be misleading to assume that non-volatility alone makes a material “green.” A more precise approach is to evaluate the entire process from start to finish. For example, both energy consumption and emissions in every step must be considered. BASF is using its own eco-efficiency analysis tool to evaluate which process from a set of possible alternatives is the most sustainable one. This analysis has been done for the ionic liquids-based BASILTM process, which was shown to be more sustainable with regard to both economics and environment. BASIL has been awarded with the eco-efficiency label.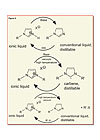
How Can Ionic Liquids Be Recycled?
Recycling of ionic liquids is easy when protonated cations are used. In this case, the ionic liquids can be switched off by deprotonation. The resulting amine or imidazole is a conventional liquid that can be distilled for recycling or purification purposes (see Figure 5).It is more difficult with the alkylated cations. Apart from purification or recycling by a liquid-liquid extraction, two principal distillation methods haven been reported: the formation of distillable carbenes [WO 01/77081] and the reversible de-alkylation of the cation [WO 01/15175; DE 10002420].
Imidazolium cations can be deprotonated by bases to form neutral carbenes. These carbenes are surprisingly stable and can be distilled. The ionic liquids can be recycled by further reaction of the carbene with an acid.
This controlled decomposition reaction even allows for a recycling or purification process of the ionic liquid. In this case, the ionic liquid is thermally cleaved. The neutral imidazole and alkylating agent are distilled off, collected and re-reacted.
How Can Ionic Liquids Be Disposed Of?
Ionic liquids are frequently reported as being non-flammable. This is only true up to the temperatures at which decomposition takes place. Some ionic liquids already start to decompose at 120°C; others are stable up to nearly 400°C. Upon decomposition, neutral and volatile molecules are formed, which can burn. This explains why ionic liquids indeed have flash points, even if they are usually much higher than 100°C. However, ionic liquids can easily be disposed of by incineration, which is usually done at temperatures of several hundred degrees Celsius. At these very high temperatures, even the toughest organic ionic liquids will give up.When Should I Think About Using Ionic Liquids?
Following are situations in which ionic liquids should be considered.- The property profile of ionic liquids could fit into an already realized application that needs improvement.
- The property profile of ionic liquids might allow results not possible with other materials.
- A problem with an application or process - or an idea for a new product or a new process - cannot sufficiently be realized with current methods.
What Prices Can Be Expected for Ionic Liquids?
Prices will mainly depend on the quantity in which specific ionic liquids are produced - most of the ionic liquids today are manufactured only in kg quantities and therefore are offered at high prices. The price will be also influenced by specific requirements on specification parameters if these make additional manufacturing steps necessary.
Which Ionic Liquid Should I Use?
Many publications mention imidazolium salts most often as preferred ionic liquid materials for several reasons. We find that imidazolium salts offer many advantages, therefore we set a focus on these. Nevertheless, with each application we evaluate, we also check other opportunities, hoping to identify the optimal ionic liquid demonstrating the best fit between the properties and the requirements of the desired application.The discussion on potential prices for ionic liquids is often limited to statements like “imidazolium salts are expensive” and “ammonium salts are cheap.” We do not see a basis for these statements, as we have not seen big differences in our cost calculations for ionic liquids with different cations. However, the anion should not be underestimated, as in many cases the anion will define the performance of the ionic liquid as well as the price (such as in electrochemical applications, where often fluorinated anions are required).
For more information on ionic liquids, contact BASF Intermediates at +49 (0)621 60-49046, fax +49 (0)621 60-42525 or visit www.basf.com.
Links
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!




