Improved Rheological Profile and Stability of 2K Waterborne Polychloroprene Dispersion Adhesives
Microfibrillated cellulose (MFC) offers multiple benefits in waterborne polychloroprene adhesive formulations.

Polychloroprene has a long history in the world of adhesives. It has been the primary choice when fast, high-strength, and permanent bonds are needed in variety of applications, from shoe soles to foam bonding. Historically, polychloroprene adhesives have been solvent borne, but environmental and occupational safety reasons have recently caused a shift toward waterborne polychloroprene adhesives. One of the challenges in waterborne polychloroprene dispersion adhesives has been related to the thickening of these high pH systems, as well as the storage stability.
Typical thickeners used for waterborne polychloroprene dispersions include methylcellulose, carboxymethylcellulose, and acrylate thickeners. The purpose of the thickener is to increase the viscosity of the finished adhesive to provide enough sag resistance, as well as to control the level of penetration of the adhesive into the substrate (e.g., in the case of foam bonding). Thickeners are also added in order to prevent the settling of pigments, fillers, or metal oxides such as zinc oxide.
Microfibrillated Cellulose
Microfibrillated cellulose (MFC) was first introduced back in the eighties.1 During the following years, its potential and possible uses have been reported extensively, ranging from uses as a thickener in paints to an oxygen barrier in food packaging.2 Until the beginning of 2010, the research and small-scale production of MFC was conducted mainly at universities and small pilot facilities. The world’s first commercial MFC production facility opened in 2016, producing 1,000 dry MT.3
MFC is a renewable, bio-based, value-added, and multifunctional product made of cellulose consisting of fibrils with lateral dimensions in the nanoscale and lengths up to micron scale. The fibrils have the ability to form stable three-dimensional networks in adhesives and sealants, leading to excellent flow and anti-sag properties. The secret behind these properties is the extremely large surface area, combined with numerous hydroxyl groups on the surface of the fibrils.
The main characteristic of this fibril structure resembles a hybrid of water-soluble polymers and insoluble additives. MFC has a high viscosity at low shear, provides yield stress, is shear thinning, and has a high water holding capacity. The insoluble nature of the cellulose fibrils also leads to excellent stability and compatibility. MFC is stable at a wide pH range (1-13) and is compatible with common water miscible organic solvents, as well as with surfactants. The viscosity of MFC is independent of the temperature, giving the same performance regardless of the surrounding conditions.
Compared to common polymer solutions in water, the rheological properties of MFC suspensions are unique. The viscosity does not reach a plateau at low shear as it normally does in polymer solutions. This can be seen in Figure 1, which shows that the viscosity of the MFC suspension increases as the shear rate decreases.
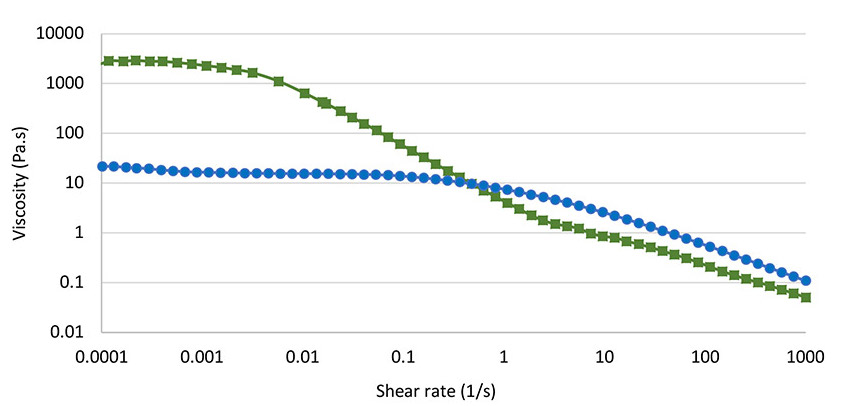 Figure 1. Flow curves for 1.0% (w/w) of MFC (square) and HEC (dot).
Figure 1. Flow curves for 1.0% (w/w) of MFC (square) and HEC (dot).
Instead, the viscosity of hydroxyethyl cellulose (HEC) solution reaches a plateau around the shear rate of 0.1 s-1. This means that the viscosity at rest is much higher for MFC, nearly 100 times higher. This will give considerably better stability under storage, especially in terms of syneresis and settling. At higher shear rates, the HEC solution and the MFC suspension have comparable shear thinning, which helps in the pumping, spraying, mixing, and application of the formulation.
One of the interesting features of MFC is its ability to allow excellent sag resistance combined with good levelling.4 The thickening mechanism of MFC is based on the physical entanglement of nano-sized fibrils and hydrogen bonding between the hydroxyl groups of MFC and hydrogen bonding compounds in the formulation. Once shear is applied, the viscosity of the adhesive reduces rapidly due to the break-up of the network.
After shearing is stopped, the network buildup is delayed due to the interference of the other hydrogen bonding components in the formulation, allowing time for the levelling to take place. This MFC network is not sensitive to changes in pH or temperature, the presence of surfactants, or other associative compounds, leading to a robust system.
Rheology Modifier for Polychloroprene Dispersions
MFC is typically available commercially either as a 2 wt% dispersion or 10 wt% paste in water. Typical dosage levels are between 5-15 wt% and 1-5 wt%, respectively. The 2 wt% dispersion can be readily added under low shear into 1K and 2K polychloroprene, but the downside is the dilution of the adhesive due to the high amount of water introduced.
The 10 wt% paste requires shear (tip speed > 6 m/s) in order to be activated, which causes challenges with the shear-sensitive waterborne polychloroprene. One of the options is to firstly disperse the 10 wt% MFC paste into a shear stable product, such as colloidal silica.5 The shear stability of waterborne polychloroprene dispersions depend on pH. They are typically stable at higher pH (> 11), allowing high-shear dispersion of compounds.
In the present study, the potential as well as compatibility of 10 wt% MFC as a rheology modifier for 2K waterborne polychloroprene dispersion adhesives was evaluated in terms of rheological and adhesive properties.
Experimental Materials and Methods
Commercially available MFC was used in this study, containing 10 wt% MFC, in water.* Two different commercial anionic polychloroprene dispersions were used, with pH 11.5 and pH 12, respectively. The MFC was dispersed into the polychloroprene dispersion under medium shear with a Dispermat® (60 mm blade/2,000 rpm) for 15 min, together with a defoamer.
After mixing, the formulations were filtered through a 200 μm stainless steel mesh filter. The rheology was measured with a Brookfield viscometer (spindle 1/30 rpm), as well as with an Anton Paar rheometer using a bob and cup measuring geometry.
Three different dosage levels of 10 wt% MFC were applied into two different commercially available polychloroprene dispersions (see Table 1). The pH of the polychloroprene dispersions was around 12. At this pH level, the dispersions are shear stable. Once MFC has been dispersed into the polychloroprene, the pH of the system can be lowered if needed.
| Dosage of 10 w% MFC (w%) | Polychloroprene 1 (mPa·s) | Polychloroprene 2 (mPa·s) |
|---|---|---|
| 0 | 27 | 28 |
| 2 | 160 | 137 |
| 4 | 364 | 461 |
| 6 | 761 | 904 |
When measuring the viscosity of polychloroprene dispersions containing MFC with a Brookfield viscometer, modest thickening can be observed (see Table 1). Due to the high shear thinning of MFC, the Brookfield is not suitable for measuring the low shear viscosity of MFC-based formulations.
Figure 2 shows the strong impact of MFC to the low shear viscosity of polychloroprene dispersions when measured with the rheometer. Depending on the dosage, the low shear viscosity varies between 10-200 Pa·s, whereas the viscosity at a shear rate of 1,000 s-1 is round 20-60 mPa·s. It is commonly known that in order to prevent sagging, the low shear viscosity should be around 25-50 Pa·s.6
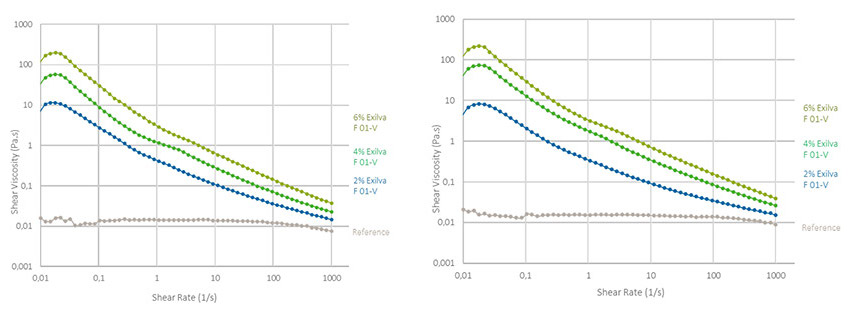
Figure 2. Shear curves of polychloroprene dispersions with varying amounts of MFC, with Polychloroprene 1 on the left and Polychloroprene 2 on the right.
The high viscosity at low shear area can also be seen by dip testing. As shown in Figure 3, wooden sticks were dipped into polychloroprene dispersion with and without MFC. The formulation containing MFC formed a uniform layer on the surface of the wooden stick, followed by sagging or dripping.

Figure 3. Dipping test using wooden sticks with neat polychloroprene dispersion on left and with MFC on the right.
Summary
MFC shows excellent compatibility with high pH (> 12) waterborne polychloroprene dispersion systems. The high shear thinning enables spraying of the product, whereas the high viscosity at rest stabilizes the formulation during storage and transport. The viscosity of the polychloroprene formulation can be adjusted by the amount and grade of MFC.
For more information, contact the author at otto.soidinsalo@borregaard.com or visit www.borregaard.com.
*Exilva® F 01-V, manufactured by Borregaard
Note: Opening image courtesy of Sandralise via iStock / Getty Images Plus on GettyImages.com.
References
1. A.F. Turbak, F.W. Snyder, and K.R. Sandberg, U.S. Patent 4,500,546, 1982.
2. D. Klemm, F. Kramer, S. Moritz, T. Lindström, M. Ankerfors, D. Gray, and A. Dorris, “Nanocelluloses: A New Family of Nature-Based Materials,” Angewandte Chemie Int. Ed., 2011, 24, 5438.
3. Sustainable microfibrillated cellulose that improves rheology and stability, www.exilva.com.
4. O. Soidinsalo, S. Holtan, A. Moosavifar, “Microfibrillated Cellulose A Novel and Renewable Multifunctional Additive for Waterborne Coatings,” Coatings World, June 2019.
5. T. Marwedel, “How to use and understand cellulose fibrils? The practical experience from Covestro,” June 26, 2018, www.exilva.com/blog/how-is-it-acutally-to-use-cellulose-fibrils-hear-covestros-experience.
6. S. Wu, “Rheology of high solid coatings. I. Analysis of sagging and slumping,” J. Appl. Polym. Sci., 1978, 22, 2769-2783
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!




