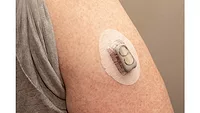
Selecting and Testing Skin Adhesives for Medical Fixation Devices
The development of adhesives and substrates for wearable medical devices is challenging and requires careful consideration due to the nature of human skin and the characteristics of the specific device.

The wearable medical device market is growing rapidly, driven by demand for innovative products that offer personalized care such as activity monitoring, point-of-care diagnostics, and dispensing (insulin, medications, electrolytes, etc.). These devices are quickly becoming a valuable part of many care plans, as they lessen the patient’s time in medical facilities—which has been critically important throughout the COVID-19 pandemic—and reduce healthcare costs.
Wearable medical devices must be stable, safe for skin contact, and unobtrusive, increasing the need for high-performing adhesives for device fixation. The ability to properly adhere to the skin is a key factor for an effectively functioning device. The adhesives used must perform consistently and attach to the body for continuous and long-term durations. Therefore, the selection and development of adhesives and substrates for wearable medical devices is challenging and requires careful consideration due to the complex and varying nature of human skin and the characteristics of the specific wearable device, such as its size, weight, flexibility, and application purpose.
Adhesive Selection
While proper screening and testing to choose the optimal wearable adhesive may seem time and labor intensive, it can ultimately save time and money by reducing risk in the long run. Moving forward with an existing adhesive carrier substrate without careful evidence might lead to failure at the clinical stage because of improper device adhesion.
The requirements for the device and its adhesive must be defined in order to identify the adhesive that is the best choice for a particular wearable device. Factors to consider include:
- Targeted users
- Application site
- Wear duration for individual application and number of expected reapplications
- Condition of use (at rest or active patient)
- Size and weight of the device
- How the device will be attached to the adhesive patch
- Whether the device will cause some occlusion of the patch or allow sweat to pass through the patch
Substrate Selection
Generally, four main types of substrates are key contributors to the development of medical-grade adhesives. The selection ultimately depends on cost, application, and performance. Substrate types include foam backing, film backing, nonwovens, and others.
Foam Backing
Foam backing is typically easy to handle and offers high breathability and excellent conformability. It can also provide cushioning.
Film Backing
Breathability ranges from low to very high with film backings. They provide great clarity if required, as well as excellent conformability (though they can be challenging to apply). Film backings can act as a bacterial barrier and are easy to print.
Nonwovens
Nonwovens feature high breathability and are often used to weld to other plastic materials. They are easy to handle, apply, and convert. Elastic variants are available.
Others
Additional backing types include cloth substrates, paper substrates, and aluminum foils (microtiter plate market).
Recommended Adhesive Testing
The first step in testing adhesives for medical devices is prototype screening. To save time during development, the screening process of the possible solutions is fundamental. An effective combination of a few in vitro and in vivo tests can drastically accelerate the development cycle.
Base characteristics of the adhesive are moisture vapor transmission rate (MVTR), peel, tack, and shear. All of these properties can be effectively screened in vitro. The goal is to compare adhesive options to one another in terms of tack, peel, and MVTR. The results will drive informed decisions on the adhesive selection and facilitate a new selection in the event the first choice of adhesive does not perform as expected in a healthy volunteer study.
The next step is to classify the adhesive prototypes from the highest to the lowest tack, peel, and MVTR. Next, the appropriate carrier will need to be selected using flexibility, durability, weldability, and breathability requirements.
Based on the classification of the adhesive, it is recommended that a study be conducted on healthy volunteers with the wearable device itself (or a surrogate that has similar weight, shape, and thickness) at the application site. To perform human testing, all biocompatibility testing, such as cytotoxicity, skin irritation, and sensitization, needs to be completed prior to the evaluation.

Selecting the Coating Method
After the medical device manufacturer has worked closely with its development partner to select the correct adhesive and backing for the device/application, technologies or chemistries should also be fine-tuned to meet a product’s unique requirements. The factors that need to be considered when selecting the optimal coating method for medical device fixation are numerous and not just application based.
Functional needs that should be considered include low pain upon removal (gentle), the need to stay in place (adhesion level), long or short wear time, sticking to broken or intact skin, and breathability. Additional questions to ask include: Does the medical device need to be a bacterial barrier or even antimicrobial? Does the material need to be stretchable or rigid? In addition to the type of substrate (e.g., film, foam, fabric, nonwoven), the type of release liner needs to be determined to prevent the adhesive from bonding to other items.
Application and packaging needs include ease of identification (for the healthcare professional applying the device), ease of handling and application, easy-to-open packaging, easy-to-understand application instructions, suitable packaging for product shelf life, and so on. The final device application is also an important consideration (e.g., transdermal drug delivery, device fixation, wearable monitoring devices such as ECGs or continuous glucose sensors).
It is also vitally important to consider a range of regulatory issues. Factors to determine include requirements regarding biocompatibility, California Prop 65, heavy metals, phthalates, BPA, free of animal derivatives and latex, and more.
Additional general considerations include the type of sterilization to be used on the device, shelf-life/storage stability, and cost.
Coating Techniques for Medical-Grade Adhesives
Each adhesive coating company has its own special techniques for coating the variety of adhesive technologies. The main criteria are machine and coating head cleanliness, as well as the elimination and reduction, wherever possible, of the risks of contamination from the environment and other manufacturing processes.
The substrate and type of coating are also important aspects when coating any of these adhesives; although most adhesives are coated as a thin continuous layer, the adhesives could be pattern coated if required. The adhesive could also be applied to the substrate either by direct coating or indirect (transfer) coating. When performing transfer coating, compatibility and wet-out of the carrier are important aspects to factor into the adhesive product design.
Depending on the adhesive type and coat weight, different processing temperatures are required for drying and curing processes. For example, acrylic adhesives can be dispersed in solvents or in water (emulsion acrylic). They might be coated via the same technique (knife over roll or reverse roll); however, they will need different drying ovens and safety measures.
For solvent adhesive systems, the lower explosive limits (LELs) need to be controlled, as the solvents are highly volatile and dangerous if not handled safely. Water-based adhesives do not have this issue; however, care must be taken with the type of drying ovens used or the machinery can start to rust and subsequently contaminate the adhesive.
In 100% solid systems, the adhesive is formed by a chemical reaction between two or more components. With these types of adhesives, the reaction is initiated by some other means (e.g., the addition of a crosslinking agent or use of a UV or radiation curing system), and thermal ovens can be used to accelerate the crosslinking process. In these systems, the adhesive will have a pot life once the reaction is started; extra care and other techniques should be utilized to ensure product consistency.
Recommended Testing to Assess Coating Performance
Depending on customer requirements, various tests may be required to check the adhesive’s suitability for the application. Peel adhesion tests, among others, are commonly used.
The standard testing method for peel adhesion is performed at either a 90˚ or 180˚ angle. The specific substrate is generally mirror-finish stainless steel, glass, or a defined plastic sheet or film.
Other tests may be performed at the design stage rather than as ongoing checks. These include dynamic and static shear adhesion tests, such as tack tests to see how quickly the adhesive sticks to the substrate. As a generality, if the adhesive has been formulated correctly, applied at the correct coat weight, and cured at the right temperature, the performance will be within the required specification.
Partner Up
When selecting the proper skin adhesive and manufacturing partner to develop a wearable medical device, it is important to ensure that the manufacturer understands the end-user application and has extensive experience in testing and delivering medical device securements that truly meet customer requirements.
For more information, visit www.scapahealthcare.com.
Note: Images courtesy of Scapa Healthcare.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!