Performance Elastomers from Renewable Resources
Elastomers produced with a new renewable monomer are helping create new market value while offering sustainability benefits.

Elastomer products are commonly produced using isoprene, butadiene, and styrene. A new renewable monomer called β-farnesene has been used to develop unique polymers, such as liquid farnesene rubber (LFR) and a proprietary bio-based styrenic elastomer.*
LFR provides fast crosslinking kinetics regardless of the curing agent and brings advantageous properties to rubber products such as tires and automotive sealants. The bio-based styrenic elastomer exhibits low hardness, high flowability, and damping properties.
Liquid Rubber Characteristics
This liquid rubber is a low-molecular-weight polydiene. The molecular weight is designed between a typical solid rubber and plasticizer, as shown in Figure 1. Termed a “reactive plasticizer,”1 the liquid rubber has characteristics of both rubbers and plasticizers, namely co-vulcanizability with solid rubbers and excellent plasticizing effects.

Figure 1. Molecular weight of liquid rubber.
Figure 2 illustrates the molecular structure of trans-β-farnesene. The circled part is trans-β-farnesene’s conjugated diene head group, which enables it to be polymerized by anionic, radical, cationic, and coordination polymerizations.2 The squared part in Figure 2 differentiates trans-β-farnesene from petroleum-sourced monomers like isoprene and butadiene. The long chain branched tail helps lower viscosities and increase reactivity.
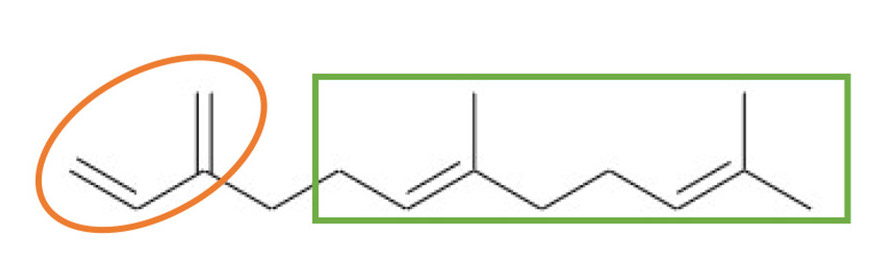
Figure 2. Structure of trans-β-farnesene.
Rubber-based automotive sealants are common in the transportation market. Several materials, such as epoxy resins, acrylic resins, urethane rubbers, and diene rubbers, are used in these sealants and adhesives. Sulfur-cured diene rubbers are often used in anti-flutter and body sealants due to their flexibility and elasticity. However, peroxide curing is becoming the preferable curing system due to the transition of pre-coating agents from zinc phosphate to zirconium oxide. Zinc phosphate coating has been widely used as an iron surface treatment.3 This coating serves two primary purposes: to protect vehicle bodies from corrosion and to promote adhesion between the paint and metal.
Although zinc phosphate coating is the most common treatment in the automotive industry, its byproduct of zinc phosphate sludge (and its disposal) have long been an environmental issue. Recently, new coating systems using zirconium oxide as an alternative to zinc phosphate have been developed. However, zirconium oxide coatings are relatively thinner and therefore weaker against sulfur corrosion. To offset this issue, organic peroxide-curable rubber formulations may be preferable to zirconium oxide coating systems.
Although diene rubbers can be crosslinked with both sulfur and peroxide, they show different curing kinetics due to their microstructures. Curing kinetics of various liquid rubbers in each curative package are shown in Figures 3 (sulfur) and 4 (peroxide). In the sulfur system, polybutadiene, having higher 1,4-structure (1,4-LBR), exhibited faster curing than polybutadiene with higher 1,2-structure (1,2-LBR). On the other hand, 1,2-LBR exhibited faster curing in the peroxide system. This may be due to the different chemical reaction processes between sulfur and peroxide crosslinking.
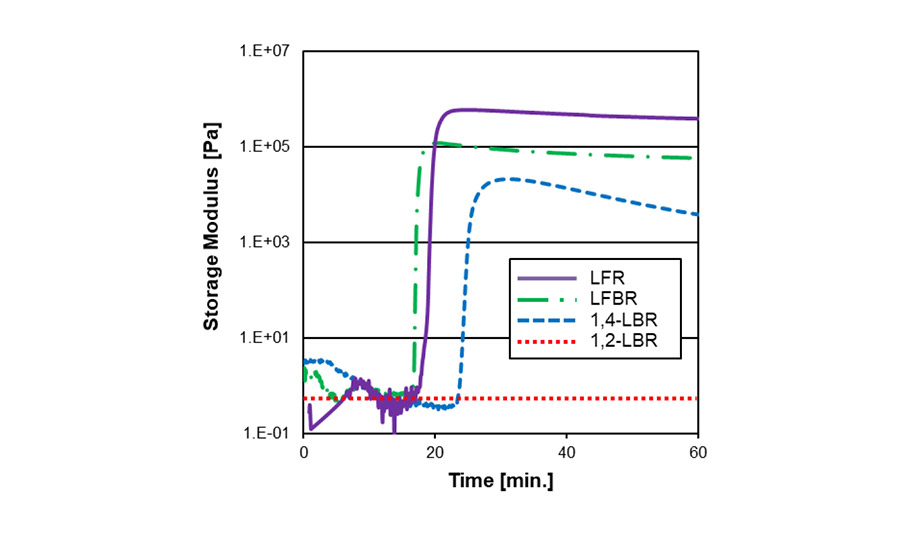
Figure 3. Crosslinking kinetics in sulfur curing.
LFR and farnesene-butadiene copolymer (LFBR) showed fast crosslinking kinetics with both sulfur and peroxide packages. It is assumed that the high reactivity of the branched side chain may contribute to the fast crosslinking kinetics.
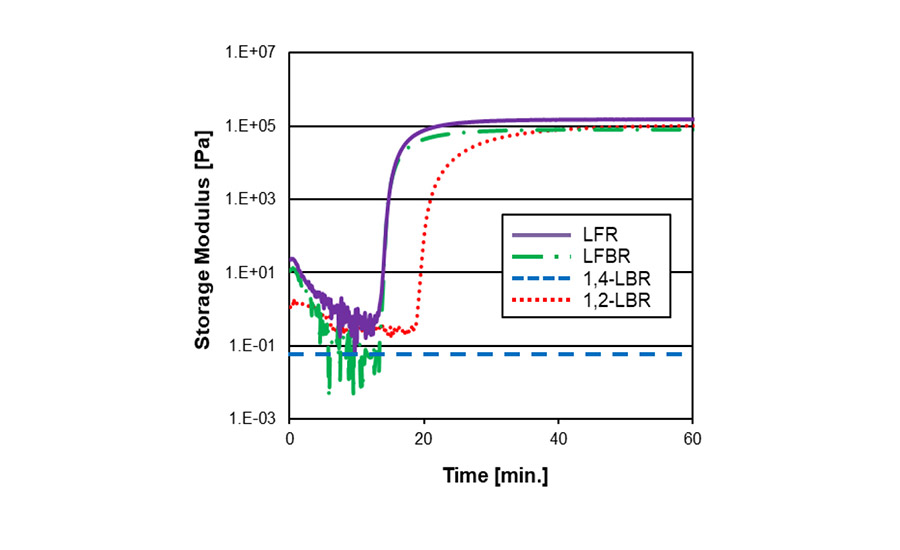
Figure 4. Crosslinking kinetics in peroxide curing.
Renewable Styrenic Elastomer
Styrenic elastomers, which are a type of thermoplastic elastomer, consist of one or more polystyrene blocks as hard segments and a polybutadiene and/or polyisoprene block as soft segments. This kind of polymer exhibits rubber-like properties since the hard segments act as physical crosslink points below the glass transition temperature (Tg), yet they flow and process at temperatures higher than the Tg.
When the polydiene block is hydrogenated, it is called a hydrogenated styrenic elastomer or hydrogenated styrenic block copolymer (HSBC). HSBCs have many advantages in comparison to non-hydrogenated styrenic copolymers, including: higher tensile strength; better heat, weather, and ozone resistance; and better compatibility with polyolefins.
The new renewable styrenic elastomer contains a hydrogenated trans-β-farnesene block as a soft segment. The highly condensed branch structure brought novel characteristics to the elastomer (see Table 1). The renewable styrenic elastomer typically shows lower hardness, higher flowability (MFR), and better compression set properties.
| Header 1 | A | B | C |
|---|---|---|---|
| Hardness (Type A) | 8 | 21 | 25 |
| MFR (230°C, 10 kg; g/10 min) | 55 | 2.1 | >700 |
| 100% modulus (MPa)1 | 0.2 | 0.2 | 0.3 |
| Tensile strength (%)1 | 4.8 | 4.6 | 5.8 |
| Elongation (%)1 | 820 | 550 | 900 |
| Compression set (70°C; %) | 35 | 21 | 100 |
| ODT (°C)2 | 330 | >350 | 210 |
| Bio-based content (%)3 | 80 | 68 | 48 |
1. Compression molded sheet of 1 mm thickness
2. Order-disorder transition temperature
3. Calculated by method developed from ASTM D6866/feeding ratio of farnesene
*Note: SEPTON™ BIO-series, A=SF902; B=SF903; C=SF904
Sustainability Contributions
Liquid farnesene rubber and the renewable styrenic elastomer represent pioneering technology that improves the environment and enhances quality of life. Life cycle assessments of these materials have been undertaken to calculate their relative greenhouse gas (GHG) emissions. The results revealed that the styrenic elastomer is expected to reduce GHG emissions by 33% in comparison to a similar, conventionally produced material. In addition, LFR is expected to reduce emissions by 108% in comparison to a similar liquid isoprene rubber. It is clear that these materials will help to create new market value due to their unique chemical structure and the reduction of GHG emissions.
For more information, visit www.kuraray.com.
*SEPTON™ BIO-series, developed by Kuraray
Editor’s note: This article is based on a paper given at the Fall 200th Technical Meeting of the Rubber Division of the American Chemical Society, Pittsburg, Pa., October 5-7, 2021.
Note: Opening image courtesy of hirun via iStock / Getty Images Plus on GettyImages.com.
References
1. M. Maeda, R. Böhm, “Liquid Rubber (LIR), a crosslinkable plasticizer,” RFP-Rubber Fibres Plastics International, 3, 152, 2009.
2. S.J. Schofer, D.J. McPhee, N. Moriguchi, Y. Yamana, B.K. Chapman, K. Hirata, Y. Uehara, “Biofene, a renewable elastomer materials with novel properties: Polymer development, characterization and use,” Rubber World, 25, August 2014.
3. M. Doerre, L. Hibbitts, G. Patrick, N.K. Akafuah, “Advances in Automotive Conversion Coatings during Pretreatment of the Body Structure,” Coatings, 8, 405, 2018
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!





