Diazirine-Based Adhesives and Primers Facilitate Bonding to Low-Surface-Energy Plastics
A new suite of reagents enables bonding of low-surface-energy materials by employing a novel mechanism achieved by using diazirine groups.

Low-surface-energy plastics are ubiquitous in modern society. Various forms of polyethylene (including LDPE, LLDPE, HDPE, UHMWPE, PEX, etc.) and polypropylene (BOPP and CPP) together constitute the most abundantly produced polyolefins on Earth,1 and these polymers are highly valued for their combination of good mechanical strength, low density, and—most importantly—low cost.2,3 As a result, they are used in everything from automotive manufacturing and construction to consumer products, surgical implants, and product packaging.4–6
Challenges to Bonding
Unfortunately, adhesive bonding of these low-surface-energy materials remains a significant challenge. Traditional adhesives work through a combination of hydrogen bonding interactions (between polar groups in the adhesive layer and those on the substrate surface), van der Waals forces, and mechanical interlocking of polymer chains.7,8 Because commodity plastics like polyethylene and polypropylene are formed from nonpolar carbon–hydrogen and carbon–carbon bonds, they cannot participate in hydrogen bonding interactions with traditional bonding agents. As a result, users of glues, epoxies, and other adhesives observe significant adhesion failure, where the applied adhesive detaches from the polymer surface.9 While it is possible to enhance the surface energy of commodity polymers through oxidation (either by chemical means, or else through plasma treatment, corona discharge, or similar high-energy processes), such methods are costly and labor intensive, and can damage the polymer substrate.10–13
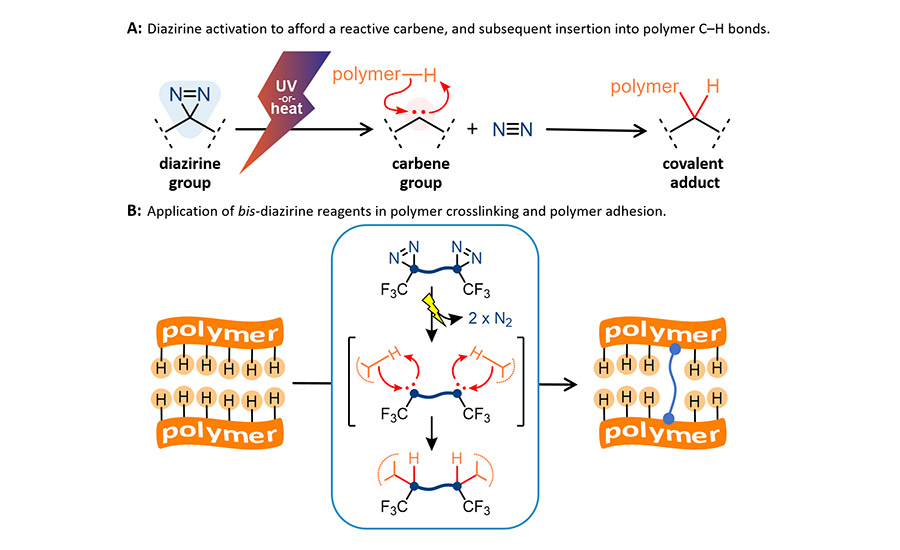
Mechanism of diazirine activation, and use of diazirines in polymer applications.
A Novel Mechanism
To address the above limitations, the Wulff Research Group at the University of Victoria, together with XlynX Materials, recently introduced a suite of new reagents that can achieve bonding to low-surface-energy materials through a wholly novel mechanism.14–22 The team makes use of diazirine groups: small chemical motifs consisting of a strained three-membered ring that incorporates a single carbon atom attached to two doubly bound nitrogen atoms (Figure 1A). When activated with heat or light, the diazirine groups expel nitrogen gas to generate highly reactive functional groups called carbenes. These carbenes can then engage in rapid insertion reactions with any nearby carbon–hydrogen (C–H), oxygen–hydrogen (O–H), or nitrogen–hydrogen (N–H) bonds. Because almost every commodity polymer (except certain fluoropolymers) contains these bonds (indeed, the surface of any given polyethylene or polypropylene chain is nothing but C–H bonds), these diazirine-containing reagents provide an easy and fast way to bond (or functionalize) the surface of virtually any polymer, whether or not it possesses a low surface energy.
XlynX Materials now offers two lines of products that allow low-surface-energy polymers to be conveniently bonded. First, the BondLynx© series of molecular adhesives comprises small molecules that incorporate two or more diazirine groups into their structures (Figure 1B). These can simply be applied between two polymer surfaces then activated by modest heat (>110 °C or >80 °C, depending on the specific product) or UV light (generally 365 nm, but up to 405 nm for some products now available) to generate carbenes that react non-specifically with the two polymer surfaces. The result is a strong covalent bond, even for surfaces that would be challenging to join by traditional means. Adhesive forces of up to 5 MPa are achievable for HDPE–BondLynx–HDPE lap-shear samples, without any need for preparation of the polymer surface and without any extensive formulation of the reagent.18 The molecular adhesive is simply dispersed in a convenient solvent, painted onto the polymer surface, and allowed to completely dry before the lap-shear ”sandwich” is assembled. It is important to remove any dispersing solvent completely prior to activation, since carbenes will preferentially react with residual solvent. Dissimilar surfaces (e.g HDPE–polypropylene or HDPE–UHMWPE) are just as easy to bond, since at a chemical level, each surface is merely a collection of C–H bonds.
The reagents are useful for a broad array of applications; however, they do come with two constraints. First, because any given BondLynx molecule is very small (10–20 Å; 0.1–0.2 nm in length), the polymer surfaces need to be sufficiently smooth for the crosslinking molecule to bridge between the polymer layers. Second, in cases where photochemical activation is preferred over thermal excitation (the former method can be much faster than the latter if a sufficiently powerful light source is available), photons need to be able to penetrate through at least one of the substrate polymers to reach the adhesive layer.
Broadened Utility
To circumvent these constraints and to broaden the utility of its products, XlynX recently introduced its second line of products, a family of polymeric diazirines called PlastiLynx©. These new reagents are comprised of polyamine backbones that are covalently linked to diazirine groups. While these reagents can be directly employed as covalent adhesives (analogous to BondLynx) they are most optimally deployed as primers for use with existing commodity adhesives (e.g. polyurethanes, cyanoacrylates, or polyurethanes) that are chosen for their specific properties (cost, curing time, mechanical toughness, etc.).
To use a polymeric diazirine as a primer (Figure 2A), the user paints a solution of the reagent on the desired polymer surface. The dispersing solvent (which can be water if required for regulatory purposes) is then allowed to fully evaporate—once again this is important for the success of the product—and the constituent diazirine groups are activated with 365 nm light (or longer wavelengths depending on the specific polymeric diazirine product). Thermal activation can be used in place of photochemical curing, if needed, but this tends to degrade the polyamine backbone of the reagent. Once activated, the diazirine groups on PlastiLynx are converted to carbenes, and some proportion of these react with the polymer surface. The result is that the substrate polymer (e.g. polyethylene or polypropylene) becomes covalently functionalized with polar amine groups. These polar groups then readily participate in strong covalent and non-covalent interactions with a wide variety of readily available bulk adhesives. In product testing, XlynX has found that the adhesive strength of lap joints made from HDPE–PlastiLynx–polyurethane–PlastiLynx–HDPE show 950% stronger bonding than control samples made without the use of the primer (Figure 2B).
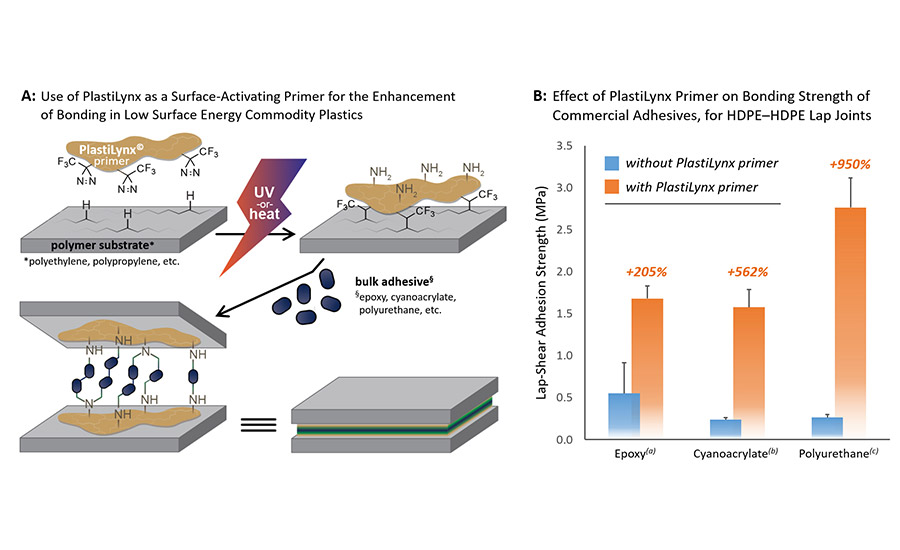
Use of PlastiLynx as an adhesion primer. Numbers in orange indicate the percentage increase in bonding strength when PlastiLynx is photocured to each HDPE bar prior to adding bulk adhesive and performing lap-shear testing. (a)West System® 105 epoxy resin containing West 205 hardener. (b)Gorilla® super glue made with 97% ethyl cyanoacrylate. (c)Lepage® PL premium polyurethane.
The primer also reacts with itself upon photochemical curing (carbenes react non-specifically with whatever C–H, O–H, or N–H bond is in closest proximity), which results in immediate conversion of the thermoplastic polyamine reagent into a hard thermoset material. This thermoplastic-to-thermoset transition allows for irreversible physical entanglement of PlastiLynx polymer chains with the chains of the substrate polymer. The cured primer can also fill any microscale cracks or crevices on the polymer surface, which in turn provides for a further increase to physical bonding.
Attaching Dyes or Pigments
As an added benefit, the amine-coated surface of the PlastiLynx-functionalized polymer can be harnessed to attach dyes or pigments (or, in principle, any other functional group). Amine-reactive dyes are particularly useful in this context, since they allow the user to easily establish a robust system of covalent linkages from the polymer surface, through the primer, to the added dye molecule. Unlike traditional dyeing methods that rely upon physical effects (and where the dye will eventually be lost from the final product), this method provides a means of attachment that resists leaching into the environment. Moreover, it is easy to encode images or logos by simply carrying out the initial photocuring step using a pre-cut mask to block the light from reaching different areas of the substrate’s surface.
BondLynx and PlastiLynx function through a mechanism of action that is completely distinct from that of any traditional adhesive, and yet work by simple topical application, much like existing glues and resins. While their use necessarily includes a curing step (this can be as short as 30 seconds), their ability to form strong covalent bonds to low-surface-energy plastics will open new doors for materials that have previously been considered resistant to adhesive bonding.
Scientists at XlynX and the University of Victoria are continuing to develop new diazirine crosslinkers for use in an even broader array of applications. They can be reached at info@xlynxmaterials.com.
Footnotes:
- R. Geyer, J. R. Jambeck, K. L. Law, “Production, use, and fate of all plastics ever made,” Science Advances 2017, 3, e1700782.
- P. Galli, G. Vecellio, “Polyolefins: The most promising large-volume materials for the 21st century,” Journal of Polymer Science Part A: Polymer Chemistry 2004, 42, 396–415.
- D. Sauter, M. Taoufik, C. Boisson, “Polyolefins, a Success Story,” Polymers 2017, 9, 185.
- C. J. Chirayil, J. Joy, H. J. Maria, I. Krupa, S. Thomas, in Polyolefin Compounds and Materials (Eds.: M. Al-Ali AlMa’adeed, I. Krupa), Springer International Publishing, Cham, 2016, pp. 265–283.
- I. Novák, A. Popelka, Z. Špitalský, I. Krupa, S. Tavman, in Polyolefin Compounds and Materials (Eds.: M. Al-Ali AlMa’adeed, I. Krupa), Springer International Publishing, Cham, 2016, pp. 181–199.
- S. J. Owonubi, S. C. Agwuncha, V. O. Fasiku, E. Mukwevho, B. A. Aderibigbe, E. R. Sadiku, D. Bezuidenhout, in Polyolefin Fibres, Elsevier, 2017, pp. 517–538.
- In Adhesives Technology Handbook, Elsevier, 2009, pp. 1–19.
- A. Popelka, I. Novak, I. Krupa, in Polyolefin Compounds and Materials (Eds.: M. Al-Ali AlMa’adeed, I. Krupa), Springer International Publishing, Cham, 2016, pp. 201–230.
- D. M. Brewis, D. Briggs, “Adhesion to polyethylene and polypropylene,” Polymer 1981, 22, 7–16.
- P. Blais, D. J. Carlsson, G. W. Csullog, D. M. Wiles, “The chromic acid etching of polyolefin surfaces, and adhesive bonding,” Journal of Colloid and Interface Science 1974, 47, 636–649.
- K. T. Lee, J. M. Goddard, J. H. Hotchkiss, “Plasma modification of polyolefin surfaces,” Packaging Technology and Science 2009, 22, 139–150.
- C. M. Plummer, L. Li, Y. Chen, “The post-modification of polyolefins with emerging synthetic methods,” Polymer Chemistry 2020, 11, 6862–6872.
- I. Sutherland, R. P. Popat, D. M. Brewis, R. Calder, “Corona Discharge Treatment of Polyolefins,” The Journal of Adhesion 1994, 46, 79–88.
- M. L. Lepage, C. Simhadri, C. Liu, M. Takaffoli, L. Bi, B. Crawford, A. S. Milani, J. E. Wulff, “A broadly applicable cross-linker for aliphatic polymers containing C–H bonds,” Science 2019, 366, 875–878.
- T. J. Cuthbert, S. Ennis, S. F. Musolino, H. L. Buckley, M. Niikura, J. E. Wulff, C. Menon, “Covalent functionalization of polypropylene filters with diazirine–photosensitizer conjugates producing visible light driven virus inactivating materials,” Scientific Reports 2021, 11, 19029.
- M. L. Lepage, M. Takaffoli, C. Simhadri, R. Mandau, M. P. Gashti, R. Nazir, M. Mohseni, W. Li, C. Liu, L. Bi, G. Falck, P. Berrang, K. Golovin, A. S. Milani, G. A. DiLabio, J. E. Wulff, “Influence of Topical Cross-Linking on Mechanical and Ballistic Performance of a Woven Ultra-High-Molecular-Weight Polyethylene Fabric Used in Soft Body Armor,” ACS Applied Polymer Materials 2021, 3, 6008–6018.
- S. F. Musolino, Z. Pei, L. Bi, G. A. DiLabio, J. E. Wulff, “Structure–function relationships in aryl diazirines reveal optimal design features to maximize C–H insertion,” Chemical Science 2021, 12, 12138–12148.
- C. Simhadri, L. Bi, M. L. Lepage, M. Takaffoli, Z. Pei, S. F. Musolino, A. S. Milani, G. A. DiLabio, J. E. Wulff, “Flexible polyfluorinated bis-diazirines as molecular adhesives,” Chemical Science 2021, 12, 4147–4153.
- S. F. Musolino, M. Mahbod, R. Nazir, L. Bi, H. A. Graham, A. S. Milani, J. E. Wulff, “Electronically optimized diazirine-based polymer crosslinkers,” Polymer Chemistry 2022, 13, 3833–3839.
- S. F. Musolino, F. Shatila, G. M. O. Tieman, A. C. Masarsky, M. C. Thibodeau, J. E. Wulff, H. L. Buckley, “Light-Induced Anti-Bacterial Effect Against Staphylococcus aureus of Porphyrin Covalently Bonded to a Polyethylene Terephthalate Surface,” ACS Omega 2022, 7, 29517–29525.
- R. Nazir, L. Bi, S. F. Musolino, O. H. Margoto, K. Çelebi, C. Mobuchon, M. Takaffoli, A. S. Milani, G. Falck, J. E. Wulff, “Polyamine–Diazirine Conjugates for Use as Primers in UHMWPE–Epoxy Composite Materials,” ACS Applied Polymer Materials 2022, 4, 1728–1742.
- X. Zhao, L. Bi, B. Khatir, P. Serles, T. Filleter, J. E. Wulff, K. Golovin, “Crosslinking inert liquidlike polydimethylsiloxane brushes using bis-diazirine chemical insertion for enhanced mechanical durability,” Chemical Engineering Journal 2022, 442, 136017.
Images courtesy of XlynX Materials Inc.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!





