Fire-Retardant Adhesives
This article examines the external events that have led to greater awareness of adhesives’ performance in the event of a fire, the standards and regulations that are being imposed, and the development of new fire-resistant materials.

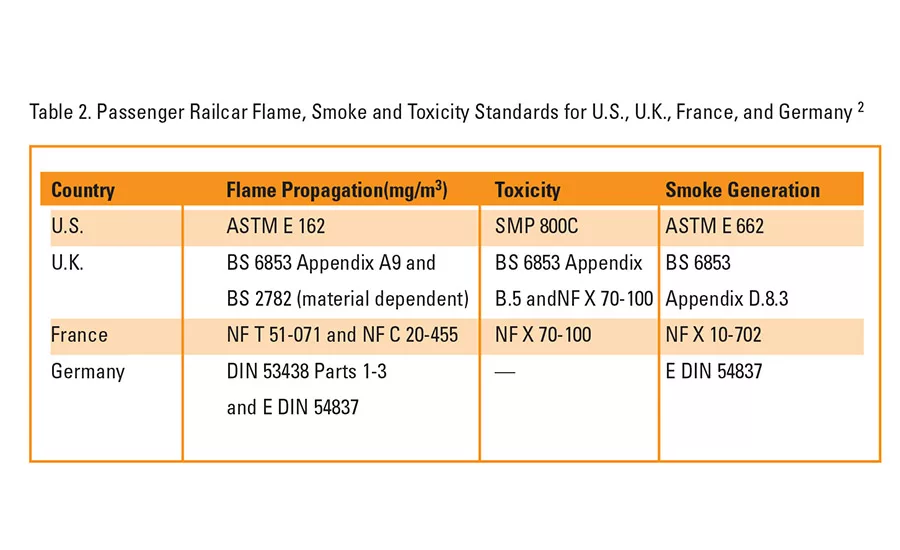
Table 2. Passenger Railcar Flame, Smoke and Toxicity Standards for U.S., UK, France, and Germany2
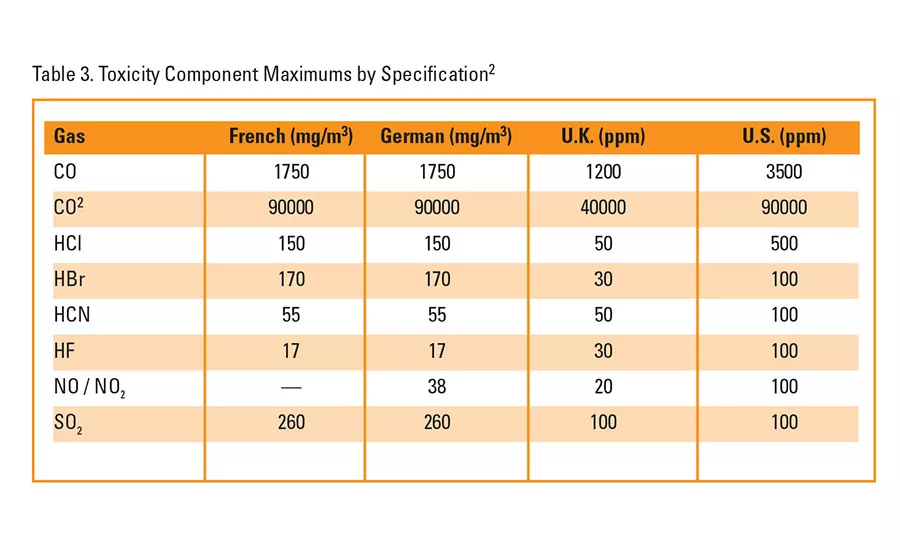
Table 3. Toxicity Component Maximums by Specification2
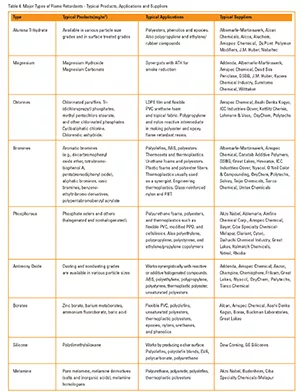
New Adhesives Requirements
Great attention is being paid to how materials perform in combating the effects of fire. This is partly due to several incidents associated with public fire outbreaks that have lead to significant damage and personal injury. They include and, unfortunately, are not limited to the following:
- The nightclub fire in Rhode Island that killed 100 people after a band’s pyrotechnics set the building’s insulation ablaze.
- The Austrian cable car fire that took 155 lives after hot oil ignited the train’s plastic flooring.
- Fires at the Manchester airport, Pipe Alpha oil rig and Kings Cross station in the UK.
In addition to a greater level of fire-safety awareness, several paths have been identified to mitigate the destruction caused when fire breaks out. These include improved structural design and the use of new fire-resisting materials. New structural designs and materials are being introduced by way of construction regulations and private architects to improve the performance of components in the case of fire. These designs are becoming accepted in industries where fire is an important concern.
Most affected are the construction and transportation (passenger rail cars, elevators, aircraft, autos, and buses) industries. One example is an increased use of composites over metal, wood and other building materials. Composites can be made to have tremendous strength, lightness and fire resistance. They are preferred over metal building materials because of their lower thermal conductivity, which inhibits the spread of high temperatures and the likelihood of fire. The regulations developed regarding these new products will likely carry over into products where fire-resistance is not generally considered (e.g., tiles, decorative laminates).
The problem with composites is that they cannot be assembled using conventional joining processes commonly used in the building and construction industry. Conventional mechanical fasteners result in high concentrations of stress, therefore adhesives are generally used. Thus, the adhesive must be equally resistant to fire and high temperatures as the flame retardant composite.
Fire resistance has always been an important requirement for materials that are used in electrical/electronic products, including those with wide public use. Printed circuit boards, flexible circuits, electrical conduits, etc., have standard flame-resistance requirements that are likely to become more rigid in the future. Many of these products use adhesives for assembly. Because of these issues, adhesives may be required to meet the following criteria.
- Resist burning
- Retain joint integrity at high temperatures
- Produce minimal smoke
- Release negligible toxic materials when heated beyond its decomposition temperature
Achieving these goals is not easy for adhesive formulators; keep in mind that the adhesive system must also provide the usual requirements of low cost, high strength, durability, and easy application. The next section will describe the state of standards and regulations being used to enforce these requirements.
Standards and Regulations
It should be recognized that emphasis regarding fire performance is always placed on a final part or product. By association, materials that make up the part or product, such as adhesives, paint, etc., are subjected to the same standards. Thus, most fire-resistance tests are designed to identify finished products that may contribute negatively to a fire. If an adhesive is used in such a product, it will be subjected to the same conditions of test and must resist burning for any significant period after removal of a source of ignition.
Standards related to fire testing generally are aimed at determining the performance of a material with respect to flame, smoke, and toxicity (FST). Several tests have been widely used to determine the resistance of materials to these conditions. These are summarized in Table 1.
The resistance of an adhesive to high temperatures in the case of a fire needs special consideration aside from the flame, smoke and toxicity effects. Often, the substrate will protect the adhesive from a fire. However, if the adhesive loosens or degrades due to the temperature of the fire, the joint can fail, causing separation of the substrate and the adhesive. If this happens, the adhesive itself becomes exposed, along with the secondary substrate. These fresh surfaces can then contribute further to the fire. No specific standard exists as of yet to measure this type of effect.
Not only does the number of standards contribute to confusion, but fire-resistant standards and regulations differ from country to country. Table 2 lists the standards for FST of passenger railcar components used in the United States, United Kingdom, France and Germany.
This disparity poses a problem in the case of components produced in countries where fire standards are less strict or nonexistent. Not only may the testing methods be different, but the limits required of the specification may also be different (see Toxicity Component Maximums by Specification, Table 3). Thus, an understanding of existing standards becomes increasingly important for companies trying to become global.
Fire retardants are in a unique position among polymeric additives - their market is sustained by some regulations, yet threatened by others. Environmental and toxicity concerns now have regulators looking with a dubious eye at the important halogenated- and antimony-based synergist flame retardants that have been developed and widely used over the years.
Take the situation with brominated epoxy resins, for example. Tetrabrominated bisphenol A epoxy resins are used in an estimated 95% of flame retardant (NEMA FR-4) printed wiring boards.3 The high bromine content (18-21%) in the molecular chain provides outstanding flame-resistant characteristics without significantly reducing the other valuable properties of the epoxy resin (strength, reactivity, viscosity, etc.).
Environmental activists, especially in Europe and Japan, tend to oppose the commercial use of halogen compounds, such as bromine containing flame retardants.
Tetrabromobisphenol A has not escaped this attention. The German Environmental Department (UBA) has issued a position paper in which they recommend phasing out tetrabromobisphenol A and claim, along with various toxicological concerns, that it has been detected in the food chain.4 There is, of course, much rebuttal regarding this position.
The Mechanism of Fire
Fire is the result of three ingredients: heat, fuel and oxygen. The heat produces flammable gases from the pyrolysis of polymer. When a certain ratio is established between these gases and oxygen, ignition of the polymer occurs. The combustion leads to a production of heat that is spread out (H1) and fed back (H2). This feedback paralyzes the polymer and keeps the combustion going.
Flame Retardants
To limit the combustion cycle one or several of the processes contributing to fire must be removed:
- Elimination of the volatile fuel, as by cooling.
- Production of a thermal barrier, as by charring, thus eliminating fuel by reducing heat transfer.
- Quenching the chain reactions in the flame, as by adding suitable radical scavengers.
Flame retardant additives do this by acting chemically and/or physically in the condensed phase or gas phase. These additives are as follows.
Char formers. Usually char formers are phosphorus compounds, which remove the carbon fuel source and provide an insulation layer against the fire’s heat.
Heat absorbers. These are usually metal hydrates such as aluminum trihydrate (ATH) or magnesium hydroxide, which remove heat by using it to evaporate water in its structure.
Flame quenchers. Usually these are bromine- or chlorine-based halogen systems, which interfere with the reactions in a flame.
Synergists. Synergists are usually antimony compounds that enhance performance of the flame quencher.
Flame retardants for polymers can also be classified as reactive (the flame retarding agent reacts chemically with the polymer to become an integral part of the molecule) or additive (nonreactive agents that are simply blended or mixed into the compound). Flame retardants can also be classified by their major chemical group (see Table 4).
There are many families of flame retardants, each with advantages and disadvantages. It is common to formulate polymers with multiple flame retardants, typically a primary flame retardant plus a synergist such as antimony oxide, to enhance overall flame resistance at the lowest cost. Several hundred different flame-retardant systems are used by the polymer industry because of these formulation practices.
Selecting a flame retardant for an adhesive systems has many ramifications, depending on the formulation being modified, the end use, how it will be processed, and the cost/performance ratio. When choosing a flame retardant, characteristics such as water extraction, particle size, viscosity, toxicity, dusting, uniformity and cost must be considered. The materials chosen to perform the function of flame retardation must not interfere with the final product’s performance. The major problem with incorporating flame retardants in adhesives is that very often a significant amount is required, and they interfere with the other properties of the adhesive and contribute to the cost.
The criteria in selecting and using the right flame retardant system for a specific adhesive formulation can be broad and somewhat complex. Care must also be taken with regard to choosing the proper test method and determining the parameters of the test. Consultation with the flame retardant suppliers is generally necessary to optimize this process.
References
- Lees, W.A.; Moulds, R.J., “Adhesives and Sealants—Their Use in the Fabrication of Fire-Resisting Components and Structures,” International Journal of Adhesion and Adhesives, 1995, pp. 95-100.
- Witthaus, E.; Hopf, R., “Don’t Get Burned by Flame Standards,” Machine Design, April 15, 2004, pp. 57-64.
- Weil, E.D.; Levchik, S., “A Review of the Current Flame Retardant Systems for Epoxy Resins,” Journal of Fire Sciences, vol. 22, January 2004, pp. 25-40.
- Leisewitz, A., et. al., Chapter V, “Summarized Substance Evaluation Tetrabromobisphenol A.,” UBA Report 204 08 542, Oko-Recherche, Frankfurt am Main, 2001.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!




